��Ŀ����
��ѧ�ķ�չ���ճ�����������أ�
��1��С�������������緹������ʾ��ΪʹӪ�����⣬�����ڽ������㽨��С����������һ����
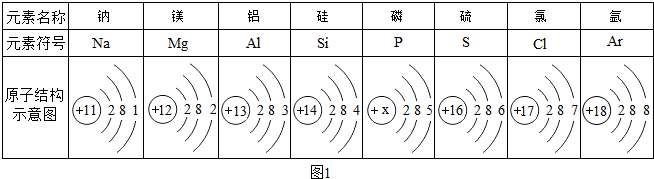
��2����������������Ԫ�غ����Ķ���ֱ��Ӱ���˵Ľ�����ȱ��ij��Ԫ�ؿ��ܵ��¼�״���״�Ԫ����
A���� B���� C��п D����
��3���·����ġ�������������������������PM2.5���ָ�꣮PM2.5��ָ������ֱ��С�ڻ����2.5�Ŀ���������彡��Ӱ�������Ҫ��Դ֮һ�ǻ�ʯȼ�ϵ�ȼ�գ����д�ʩ�ܼ���PM2.5��Ⱦ����
A��������˽�ҳ����� B������ʹ��̫������ˮ��
C��������ú�������� D��������Һ��ʯ������ȼ��
��4���������и���Ʒʹ�õIJ����У������л��ϳɲ��ϵ���
A�����ֱ��� B����ɰ�� C��ӡ���� D�����ڳ��� E���������� F����ľ
��5��������̼�ǵ�������ЧӦ����Ҫ���壬ͬʱ����Ҳ��һ�ֱ����̼����Դ����CO2��NH3�ϳ�����CO��NH2��2�ǹ̶�������CO2�ijɹ��������÷�Ӧ���ڸ��¸�ѹ�½��У�ͬʱ��ˮ���ɣ�д����Ӧ�Ļ�ѧ����ʽ ��
��6������β���г�����һ��������һ����̼��Ϊ����β���Ի�����Ӱ�죬�����ܳ���װ��ý��һ��������һ����̼�ڴ�ý�������·�����Ӧ���ɶ�����̼�ʹ�������Ҫ�ɷ�֮һ����д����Ӧ�Ļ�ѧ����ʽ ��
��1��С�������������緹������ʾ��ΪʹӪ�����⣬�����ڽ������㽨��С����������һ����
| ��ʳ | �� | �� |
| ������ | �����㡢�����Źǡ� | ���ײ����� |
A���� B���� C��п D����
��3���·����ġ�������������������������PM2.5���ָ�꣮PM2.5��ָ������ֱ��С�ڻ����2.5�Ŀ���������彡��Ӱ�������Ҫ��Դ֮һ�ǻ�ʯȼ�ϵ�ȼ�գ����д�ʩ�ܼ���PM2.5��Ⱦ����
A��������˽�ҳ����� B������ʹ��̫������ˮ��
C��������ú�������� D��������Һ��ʯ������ȼ��
��4���������и���Ʒʹ�õIJ����У������л��ϳɲ��ϵ���
A�����ֱ��� B����ɰ�� C��ӡ���� D�����ڳ��� E���������� F����ľ
��5��������̼�ǵ�������ЧӦ����Ҫ���壬ͬʱ����Ҳ��һ�ֱ����̼����Դ����CO2��NH3�ϳ�����CO��NH2��2�ǹ̶�������CO2�ijɹ��������÷�Ӧ���ڸ��¸�ѹ�½��У�ͬʱ��ˮ���ɣ�д����Ӧ�Ļ�ѧ����ʽ
��6������β���г�����һ��������һ����̼��Ϊ����β���Ի�����Ӱ�죬�����ܳ���װ��ý��һ��������һ����̼�ڴ�ý�������·�����Ӧ���ɶ�����̼�ʹ�������Ҫ�ɷ�֮һ����д����Ӧ�Ļ�ѧ����ʽ
���㣺����Ӫ���뽡��,���ο�����Ⱦ�Ĵ�ʩ,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,�ϳɲ��ϵ�ʹ�ü�����˺ͻ�����Ӱ��,�����Ԫ�������Ԫ�ض����彡������Ҫ����
ר�⣺��ѧ������
��������1���������⣬��ʳ����ȱ�ٸ���ά���ص�ʳ���Ӧ���Ӹ���ά���ص�ʳ��ݴ˽��з������
��2�����ݵ���������ܺ�ȱ��֢���з����жϣ�
��3��A��˽�ҳ�Խ�࣬ȼ�յĻ�ʯȼ��Խ�ࣻ
B��̫������ˮ������ȼ�ջ�ʯȼ�ϣ�
C��ú���ڻ�ʯȼ�ϣ�
D��Һ��ʯ����Ҳ���ڻ�ʯȼ��
��4���л��ϳɲ��ϼ�ƺϳɲ��ϣ�Ҫ�ж��Ƿ����ںϳɲ��ϣ���ץס�����������л���ϳɡ��߷��ӻ�����ݴ˳������ϵķ�����з����ж�
��5����6���������е������ҳ���Ӧ��������Լ���Ӧ������Ȼ����ݷ���ʽ����дԭ����д��ѧ����ʽ��
��2�����ݵ���������ܺ�ȱ��֢���з����жϣ�
��3��A��˽�ҳ�Խ�࣬ȼ�յĻ�ʯȼ��Խ�ࣻ
B��̫������ˮ������ȼ�ջ�ʯȼ�ϣ�
C��ú���ڻ�ʯȼ�ϣ�
D��Һ��ʯ����Ҳ���ڻ�ʯȼ��
��4���л��ϳɲ��ϼ�ƺϳɲ��ϣ�Ҫ�ж��Ƿ����ںϳɲ��ϣ���ץס�����������л���ϳɡ��߷��ӻ�����ݴ˳������ϵķ�����з����ж�
��5����6���������е������ҳ���Ӧ��������Լ���Ӧ������Ȼ����ݷ���ʽ����дԭ����д��ѧ����ʽ��
����⣺������Ҫ������Ӫ�����ʣ������ʡ����ࡢ��֬��ά���ء����κ�ˮ��������⣬���и������ۣ������������ࣻ�����㡢�����Źǡ����ײ����ຬ����֬�������ʺ����Σ���֪ʳ����ά���غ������٣���ΪʹӪ��������Ӻ�������Ҫ���主��ά���ص�ʳ��߲��༴�ɣ�
��2�����Ǻϳɼ�״�ټ��ص���ҪԪ�أ�ȱ���Ỽ��״���״�
��3��A��˽�ҳ�Խ�࣬ȼ�յĻ�ʯȼ��Խ�࣬���ɵ�С������Խ�࣬������PM2.5��Ⱦ����A���������⣻
B��̫������ˮ������ȼ�ջ�ʯȼ�ϣ��ǽྻ����Դ����������PM2.5��Ⱦ����B�������⣻
C��ú���ڻ�ʯȼ�ϣ�ȼ������С�����������PM2.5��Ⱦ����C���������⣻
D��Һ��ʯ����Ҳ���ڻ�ʯȼ�ϣ�ȼ������С�����������PM2.5��Ⱦ����D���������⣮
��ѡ��B
��4���л��ϳɲ��ϱ���߱������������˹��ϳɡ��л���߷��ӣ�D�����ڳ���E����������F����ľ�����������������л���ϳɡ��߷��ӻ�������л��ϳɲ���
��5��������̼�Ͱ����ڸ��¸�ѹ���������������غ�ˮ���ʴ�Ϊ��CO2+2NH3
CO��NH2��2+H2O��
��6��������Ŀ��Ϣ��֪����Ӧ����������Ѿ�������ע����ƽ�ͷ�Ӧ�������ʴ�Ϊ��2NO+2CO
2CO2+N2
�ʴ�Ϊ����1���߲��༴�� ��2��A ��3��B ��4��DEF
��5��CO2+2NH3
CO��NH2��2+H2O����6��2CO+2NO
N2+2CO2
��2�����Ǻϳɼ�״�ټ��ص���ҪԪ�أ�ȱ���Ỽ��״���״�
��3��A��˽�ҳ�Խ�࣬ȼ�յĻ�ʯȼ��Խ�࣬���ɵ�С������Խ�࣬������PM2.5��Ⱦ����A���������⣻
B��̫������ˮ������ȼ�ջ�ʯȼ�ϣ��ǽྻ����Դ����������PM2.5��Ⱦ����B�������⣻
C��ú���ڻ�ʯȼ�ϣ�ȼ������С�����������PM2.5��Ⱦ����C���������⣻
D��Һ��ʯ����Ҳ���ڻ�ʯȼ�ϣ�ȼ������С�����������PM2.5��Ⱦ����D���������⣮
��ѡ��B
��4���л��ϳɲ��ϱ���߱������������˹��ϳɡ��л���߷��ӣ�D�����ڳ���E����������F����ľ�����������������л���ϳɡ��߷��ӻ�������л��ϳɲ���
��5��������̼�Ͱ����ڸ��¸�ѹ���������������غ�ˮ���ʴ�Ϊ��CO2+2NH3
| ||
��6��������Ŀ��Ϣ��֪����Ӧ����������Ѿ�������ע����ƽ�ͷ�Ӧ�������ʴ�Ϊ��2NO+2CO
| ||
�ʴ�Ϊ����1���߲��༴�� ��2��A ��3��B ��4��DEF
��5��CO2+2NH3
| ||
| ||
������������һ���ۺ��⣬����ʳƷӪ������Ԫ���Լ���ѧ����ʽ�ȼ�����֪ʶ��ע��ѧ��������������

��ϰ��ϵ�д�
 Ʒѧ˫�ž�ϵ�д�
Ʒѧ˫�ž�ϵ�д� Сѧ��ĩ���100��ϵ�д�
Сѧ��ĩ���100��ϵ�д� ��ĩ��ϰ���ϵ�д�
��ĩ��ϰ���ϵ�д� ����ѧ�䵥Ԫ������ĩר����100��ϵ�д�
����ѧ�䵥Ԫ������ĩר����100��ϵ�д� �Ƹ�360�ȶ����ܾ�ϵ�д�
�Ƹ�360�ȶ����ܾ�ϵ�д�
�����Ŀ
�������������ڻ�ѧ�仯���ǣ�������
| A������̿������ɫ���ж������� |
| B��Ǧ�ʰ��ּ�����ֽ�� |
| C���ý��ʯ�и�� |
| D��ʯī�ڿ�����ȼ�� |

 ����ʾ̼ԭ�ӣ��á�
����ʾ̼ԭ�ӣ��á� ����ʾ��ԭ�ӣ��á�
����ʾ��ԭ�ӣ��á� ����ʾ��ԭ�ӣ���ͼΪ����ת�����۹��̣��������ͼʾ�ش����⣺
����ʾ��ԭ�ӣ���ͼΪ����ת�����۹��̣��������ͼʾ�ش����⣺