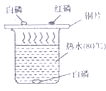
��Ŀ����
����Ŀ�������д����л�ѧ����ѧ������������أ�
��1��ɽҩ�к��м��������أ�Ƥ��մ�ϻ��������̣�����Ϊ��Ϳ�����е���ֹ����
��2��ɭ�ַ�������ʱ����������һ���������Ŀ������
��3�����dz���ϴ�Ӽ���ϴ�;��ϵ����ۣ�������Ϊϴ�Ӽ��������ܣ�
��4�����ɰ�װ���г��õĸ���������ѧʽ����
��5��������Ʒ��ʹ�õ���Ҫ�������ںϳ��л��߷��Ӳ��ϵ���������ţ���
���մɱ� �ڻ�ɳ ����ë�� ��ú̿ �ݲ������ �������� �߾ƾ�
��6����ʩ���̬����ʱ��Ҫ��������ʻ����Ա����Ч���ͣ���д�����������ʯ�һ���г�ʱ������Ӧ�Ļ�ѧ����ʽ ��
���𰸡�
��1��ʳ��
��2�������ȼ��
��3���黯
��4��CaO
��5����
��6����NH4��2SO4+Ca��OH��2![]() CaSO4+2NH3��+2H2O
CaSO4+2NH3��+2H2O
���������⣺��1��ʳ�׳����ԣ�������������ʷ�Ӧ������ɽҩ�к��м��������أ�Ƥ��մ�ϻ��������̣���Ϳ�����е�ʳ��ֹ������2��ɭ�ֻ���ʱ����������һ���������Ŀ���Ǹ����ȼ���3��ϴ�Ӽ���ϴ�;��ϵ������ǽ����۷ֽ��С���͵Σ���ˮ���ߣ��������黯ԭ��������˵��ϴ�Ӽ������黯�Ĺ��ܣ���4��ʳƷ��װ���еĸ���������ƣ���5�����������ںϳ��л��߷��Ӳ��ϣ���6������狀��������Ʒ�Ӧ��������ơ�ˮ�Ͱ�������ѧ����ʽΪ����NH4��2SO4+Ca��OH��2![]() CaSO4+2NH3��+2H2O�� ���Դ��ǣ���1��ʳ�ף���2�������ȼ���3���黯����4��CaO����5���ޣ�6����NH4��2SO4+Ca��OH��2
CaSO4+2NH3��+2H2O�� ���Դ��ǣ���1��ʳ�ף���2�������ȼ���3���黯����4��CaO����5���ޣ�6����NH4��2SO4+Ca��OH��2![]() CaSO4+2NH3��+2H2O��
CaSO4+2NH3��+2H2O��
�����㾫����������Ҫ�������кͷ�Ӧ����Ӧ�ú���д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ�����֪ʶ�㣬��Ҫ�����кͷ�Ӧ����������������κ�ˮ�ķ�Ӧ��ע�⣺a����ƽ b������ c�����Ų�����ȷ�����⣮