��Ŀ����
��Դ����������������ϢϢ��أ���ش��������⣺
��1����̫���ܡ�ʯ�͡�ˮ�ܡ�ú̿�У����ڲ���������Դ���� ��
��2��������ú¯ȡůʱӦע���ͨ�磬��Ϊú̿�����ȼ��ʱ������ж����壬�������� ��
��3��������������ԴΣ���������ܼ��š���ÿ���˵����κ���������Ҫ�������еĵ������Ϊ�������ͬ�ļ�-��������һ��Ŭ���������������������ܡ����š�д�����㽨�飺 ��
��1����̫���ܡ�ʯ�͡�ˮ�ܡ�ú̿�У����ڲ���������Դ����
��2��������ú¯ȡůʱӦע���ͨ�磬��Ϊú̿�����ȼ��ʱ������ж����壬��������
��3��������������ԴΣ���������ܼ��š���ÿ���˵����κ���������Ҫ�������еĵ������Ϊ�������ͬ�ļ�-��������һ��Ŭ���������������������ܡ����š�д�����㽨�飺
���㣺������Դ�����ࡢ��Դ�ķ���,���ο�����Ⱦ�Ĵ�ʩ,һ����̼�Ķ���
ר�⣺��ѧ����Դ
��������1�����ݿ�������Դ�Ͳ���������Դ���ص������գ�
��2��һ����̼�Ķ�����ָ������������ѪҺ�е�Ѫ�쵰��ϣ��Ӷ�ʹѪ�쵰�ײ��ܺܺõ���������ϣ�ʧȥ��Я�������������������ȱ�����������ж�����
��3�����������е����ý��ܼ�����������Щ�����ǽ�Լ��Դ��������Ĵ�ʩ����ϵ������н��
��2��һ����̼�Ķ�����ָ������������ѪҺ�е�Ѫ�쵰��ϣ��Ӷ�ʹѪ�쵰�ײ��ܺܺõ���������ϣ�ʧȥ��Я�������������������ȱ�����������ж�����
��3�����������е����ý��ܼ�����������Щ�����ǽ�Լ��Դ��������Ĵ�ʩ����ϵ������н��
����⣺��1��̫���ܡ�ʯ�͡�ˮ�ܡ�ú̿����������Դ�У�ʯ�ͺ�ú̿���ڲ���������Դ��
���ʯ�͡�ú̿��
��2��ú̿�����ȼ��ʱ�����һ����̼��һ����̼����ѪҺ�е�Ѫ�쵰��ϣ�ʹѪ�쵰��ʧȥ������ϵ�������ʹ����ȱ�����ж���
���һ����̼��
��3���ٽ��ܵĴ�ʩ�У���ʹ�������Դ������ʹ�÷��ܡ�ˮ�ܡ�̫���ܵȣ��������õ���ˮ���õ��ϰ�ߣ�
�ڼ��ŵĴ�ʩ�У���ǿ�����ŷŹ������������Ƕ����������ͨ���ߣ���������β�����ŷţ�
�ʴ����ʹ�������Դ����ǿ�����ŷŹ����ȣ�
���ʯ�͡�ú̿��
��2��ú̿�����ȼ��ʱ�����һ����̼��һ����̼����ѪҺ�е�Ѫ�쵰��ϣ�ʹѪ�쵰��ʧȥ������ϵ�������ʹ����ȱ�����ж���
���һ����̼��
��3���ٽ��ܵĴ�ʩ�У���ʹ�������Դ������ʹ�÷��ܡ�ˮ�ܡ�̫���ܵȣ��������õ���ˮ���õ��ϰ�ߣ�
�ڼ��ŵĴ�ʩ�У���ǿ�����ŷŹ������������Ƕ����������ͨ���ߣ���������β�����ŷţ�
�ʴ����ʹ�������Դ����ǿ�����ŷŹ����ȣ�
������������Ҫ������Դ�ķ��ࡢ���ʵ����ʺ���;�����ʾ��ж������ʣ����ʱӦ���������ʵ���;�������ʵ��������ʾ����ģ�

��ϰ��ϵ�д�
�����Ŀ
���и��������ڸ����������ܴ���������ǣ�������
| A���� pH=1 ����Һ�У�NaCl��Na2CO3��Na2SO4 |
| B���� pH=2 ����Һ�У�KCl��Na2SO4��HNO3 |
| C���� pH=13 ����Һ�У�BaCl��Na2CO3��NaOH |
| D���� pH=14 ����Һ�У�FeCl3��Ba��NO3��2��CaCl2 |
�����ͻ�ѧ������ʵ��Ϊ������ѧ�ƣ������йس��õ�����ʵ������˵����ȷ���ǣ�������
| A��������Һʱ�������¶ȼƴ��沣�������н��� |
| B����������ƽ����ȷ��ȡ��3.24g����ҩƷ |
| C����Ͳû��0�̶� |
| D���Թܺ��ձ��������ھƾ��ƻ�����ֱ�Ӽ��� |


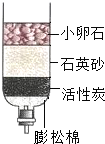 С�����òɼ����ĺ�ˮ��������-����ͼ��ʾ�ļ���ˮ�����о������Իش�
С�����òɼ����ĺ�ˮ��������-����ͼ��ʾ�ļ���ˮ�����о������Իش�