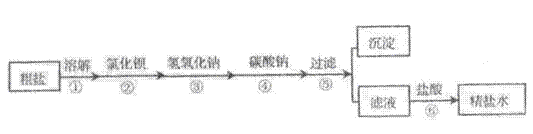
��Ŀ����
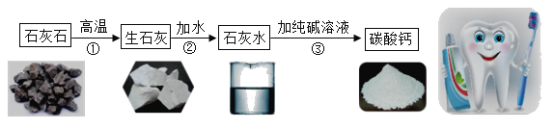
����̼��ƣ��ֳƳ���̼��ƣ����ߴ��ȵ�̼��ƣ����ܶ�С���ȸߡ�Ħ��ϵ��С�������������ԭ��֮һ�������ڸߵ���dzɫ��Ʒ�ϡ���ҵ��ͨ����ʯ��ʯ����Ҫ�ɷ�ΪCaCO3��Ϊԭ�ϣ�����ͼ��ʾ�������̽��иߴ���̼��Ƶ���ȡ���ᴿ��
����������Ϣ���Իش��������⣺
��ʯ��ʯ����;�dz��㷺�������й�˵����ȷ����_____������ѡ��
�ٿ�������ȡ������̼ �ڿ�������ȡ��ʯ�� �ۿ��������첣��
�ܿ���������������ˮ �ݿɼ���ú̿��������� ��������¯����������
A �٢ۢݢ� B �ڢܢݢ� C �٢ڢܢݢ� D ����ȫ������
�������������漰�������У��������������������_____��
���������̵IJ�����У�������ѡ������һ���Լ����ó������Ʊ����ߴ��ȵ�̼��ƣ��䷴Ӧԭ���Ļ�ѧ����ʽΪ_____��
���Ʊ��ߴ���̼���Ҫ������ʯ��ʯ��CaCO3������ʯ�ҡ�ʯ��ˮ��CaCO3����ת�����̣���Ŀ����_____��
��Сӱͬѧ��ȡ��25.0gijʯ��ʯ��Ʒ������ͼ���̽�����ģ��ʵ��̽�������ڲ��������ʯ��ˮ��μ���������������Ϊ10%��Na2CO3��Һ���ù����в������������������Na2CO3��Һ��������ϵ��ͼ��ʾ���Լ��㣺
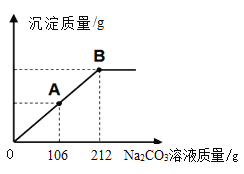
����ʵ���У�Сӱ�����Ƶ�����̼��Ƶ�������_____�������ȷ��0.1g��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д��±��У���ȥ�����������ʵķ�����ȷ���ǣ� ��
ѡ�� | ���� | �������� | ��ȥ���ʵķ��� |
A | Cu(NO3)2��Һ | AgNO3��Һ | �������������ۣ����� |
B | NaCl���� | ��ɳ | ��ˮ�ܽ⡢���ˡ����� |
C | CO2���� | CO���� | ��ȼ |
D | KNO3 ��Һ | Ba(NO3)2 | �μӹ���K2SO4��Һ������ |
A.A B.B C.C D.D


 ��
�� )�������������( )
)�������������( )