��Ŀ����
��2010?����ҹ����ϴֵ����������صĺ��֣������ڿ������ֵ�ͬʱ����Ӧ��������ˮ��������ϧˮ��Դ����ش��������⣺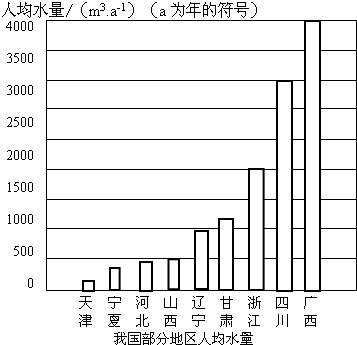
��1������������ˮ���Ǵ���ˮ������Ϊ����ˮ��ѧ���ʵ���С������
����ѡ������ȷ��ˮ������Ԫ�غ���Ԫ����ɵ�ʵ����
A��ˮ������ B��ˮ������ C��ˮ�ĵ�� D��ˮ�ľ���
��2����ͼ���ҹ����ֵ����˾�ˮ��ͼ�����п��Կ��������Ǽ���ȱˮ�ij��У������������Ͻ�Լ��ˮ����
A��ʹ�ý�ˮ��ͷ
B��������ˮϴ��
C������ࡢ�ι����ũ���スˮ
D��ʹ�ò���ϵ���ˮ�߳��ܡ���ˮ����ˮ��Ϊ165L/�εļ���ϴ�»�
��3��ʵ��������һ������MgCl2��CaCl2��Ӳˮ���±���ij��ȤС���������Ӳˮ��ʵ�鷽��������һ��������±���
��ѡ�õ�ҩƷ�У�Ca��OH��2��Һ��NaOH��Һ��Na2CO3��Һ������ˮ

��1������������ˮ���Ǵ���ˮ������Ϊ����ˮ��ѧ���ʵ���С������
ˮ����
ˮ����
������ѡ������ȷ��ˮ������Ԫ�غ���Ԫ����ɵ�ʵ����
C
C
������ţ���ͬ����A��ˮ������ B��ˮ������ C��ˮ�ĵ�� D��ˮ�ľ���
��2����ͼ���ҹ����ֵ����˾�ˮ��ͼ�����п��Կ��������Ǽ���ȱˮ�ij��У������������Ͻ�Լ��ˮ����
ABC
ABC
��A��ʹ�ý�ˮ��ͷ
B��������ˮϴ��
C������ࡢ�ι����ũ���スˮ
D��ʹ�ò���ϵ���ˮ�߳��ܡ���ˮ����ˮ��Ϊ165L/�εļ���ϴ�»�
��3��ʵ��������һ������MgCl2��CaCl2��Ӳˮ���±���ij��ȤС���������Ӳˮ��ʵ�鷽��������һ��������±���
��ѡ�õ�ҩƷ�У�Ca��OH��2��Һ��NaOH��Һ��Na2CO3��Һ������ˮ
| ʵ����� | ���� | ���ۻ�ѧ����ʽ |
| ��ȡ����Ӳˮ��Ʒ���Թ��У������еμ�Ca��OH��2��Һ��ֱ�����ٲ������� | �а�ɫ�������� | MgCl2+Ca��OH��2=Mg��OH��2��+CaCl2 |
| �����������Թ��еμ� ̼������Һ ̼������Һ ��ֱ�����ٲ������� | �а�ɫ�������� | ��ѧ����ʽ�� CaCl2+Na2CO3�T2NaCl+CaCO3�� CaCl2+Na2CO3�T2NaCl+CaCO3�� |
| �۹��� | �˳����� | �õ�������Һ |
| �������Թֱܷ�ȡ������������Һ��ԭӲˮ��Ʒ���ֱ�μ� �����ķ���ˮ �����ķ���ˮ ���� | ʢ����Һ���Թ��� ��������ĭ�϶� ��������ĭ�϶� | ֤��Ӳˮ������ |
��������1�����ݷ��ӵĶ���͵��ˮ��ʵ����ۻش�
��2���ӽ�Լ��ˮ�ķ������ǣ�
��3���ڴ����ʵķ���;����������֣�ѡ���ʵ������ʰ�Ӳˮ�е��������γ�ȥ��Ȼ����������غ㶨����д��ѧ����ʽ���ܴ�����Ӳˮ����ˮ�ķ�����ѡ�����ʻش�
��2���ӽ�Լ��ˮ�ķ������ǣ�
��3���ڴ����ʵķ���;����������֣�ѡ���ʵ������ʰ�Ӳˮ�е��������γ�ȥ��Ȼ����������غ㶨����д��ѧ����ʽ���ܴ�����Ӳˮ����ˮ�ķ�����ѡ�����ʻش�
����⣺��1��ˮ�����DZ���ˮ�Ļ�ѧ���ʵ���С���ӣ����ˮ������������������˵��ˮ��������Ԫ����ɵģ�
��2���ӽ�Լ��ˮ�ķ������ǣ�ʹ�ý�ˮ��ͷ��������ˮϴ�룬����ࡢ�ι����ũ���スˮ���ǽ�ˮ�ĺô�ʩ����ѡABC��
��3���ڵ�һ����Ӧ���������Ƴ�ȥ���Ȼ�þ���ʣ��ڶ������dz��Ȼ������ʣ����Ȼ���ʵ�ʾ��dz������ӣ�����̼������Һ��ȥ����ѧ����ʽΪ��CaCl2+Na2CO3�T2NaCl+CaCO3����
��֤��Ӳˮ�����ķ��������÷���ˮ���飬���Ƿ��ܲ�����������ĭ��
�𰸣���1��ˮ���ӣ�C
��2��ABC��
��3��
��2���ӽ�Լ��ˮ�ķ������ǣ�ʹ�ý�ˮ��ͷ��������ˮϴ�룬����ࡢ�ι����ũ���スˮ���ǽ�ˮ�ĺô�ʩ����ѡABC��
��3���ڵ�һ����Ӧ���������Ƴ�ȥ���Ȼ�þ���ʣ��ڶ������dz��Ȼ������ʣ����Ȼ���ʵ�ʾ��dz������ӣ�����̼������Һ��ȥ����ѧ����ʽΪ��CaCl2+Na2CO3�T2NaCl+CaCO3����
��֤��Ӳˮ�����ķ��������÷���ˮ���飬���Ƿ��ܲ�����������ĭ��
�𰸣���1��ˮ���ӣ�C
��2��ABC��
��3��
| ʵ����� | ���� | ���ۻ�ѧ����ʽ |
| �� | ||
| �� ̼������Һ | CaCl2+Na2CO3�T2NaCl+CaCO3�� | |
| �� | ||
| �� �����ķ���ˮ | ��������ĭ�϶� |
���������⿼���˷��ӵĶ��壬���ˮ����ˮ���������ʷ�����ᴿ�ķ�����Ӳˮ����ˮ�ļ���ȶ��֪ʶ�㣬�ۺ��ԱȽ�ǿ���Ѷ����У�

��ϰ��ϵ�д�
�����Ŀ
��2010?����±���ijͬѧ�ö��ַ����������ʵ�������������ַ�������ȷ���ǣ�������
| ||||||||||||||||||||||
��2010?����ҹ����ϴֵ����������صĺ��֣������ڿ������ֵ�ͬʱ����Ӧ��������ˮ��������ϧˮ��Դ����ش��������⣺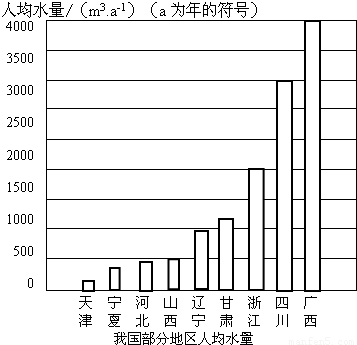
��1������������ˮ���Ǵ���ˮ������Ϊ����ˮ��ѧ���ʵ���С������______��
����ѡ������ȷ��ˮ������Ԫ�غ���Ԫ����ɵ�ʵ����______������ţ���ͬ����
A��ˮ������ B��ˮ������ C��ˮ�ĵ�� D��ˮ�ľ���
��2����ͼ���ҹ����ֵ����˾�ˮ��ͼ�����п��Կ��������Ǽ���ȱˮ�ij��У������������Ͻ�Լ��ˮ����______��
A��ʹ�ý�ˮ��ͷ
B��������ˮϴ��
C������ࡢ�ι����ũ���スˮ
D��ʹ�ò���ϵ���ˮ�߳��ܡ���ˮ����ˮ��Ϊ165L/�εļ���ϴ�»�
��3��ʵ��������һ������MgCl2��CaCl2��Ӳˮ���±���ij��ȤС���������Ӳˮ��ʵ�鷽��������һ��������±���
��ѡ�õ�ҩƷ�У�Ca��OH��2��Һ��NaOH��Һ��Na2CO3��Һ������ˮ

��1������������ˮ���Ǵ���ˮ������Ϊ����ˮ��ѧ���ʵ���С������______��
����ѡ������ȷ��ˮ������Ԫ�غ���Ԫ����ɵ�ʵ����______������ţ���ͬ����
A��ˮ������ B��ˮ������ C��ˮ�ĵ�� D��ˮ�ľ���
��2����ͼ���ҹ����ֵ����˾�ˮ��ͼ�����п��Կ��������Ǽ���ȱˮ�ij��У������������Ͻ�Լ��ˮ����______��
A��ʹ�ý�ˮ��ͷ
B��������ˮϴ��
C������ࡢ�ι����ũ���スˮ
D��ʹ�ò���ϵ���ˮ�߳��ܡ���ˮ����ˮ��Ϊ165L/�εļ���ϴ�»�
��3��ʵ��������һ������MgCl2��CaCl2��Ӳˮ���±���ij��ȤС���������Ӳˮ��ʵ�鷽��������һ��������±���
��ѡ�õ�ҩƷ�У�Ca��OH��2��Һ��NaOH��Һ��Na2CO3��Һ������ˮ
| ʵ����� | ���� | ���ۻ�ѧ����ʽ |
| ��ȡ����Ӳˮ��Ʒ���Թ��У������еμ�Ca��OH��2��Һ��ֱ�����ٲ������� | �а�ɫ�������� | MgCl2+Ca��OH��2=Mg��OH��2��+CaCl2 |
| �����������Թ��еμ�______��ֱ�����ٲ������� | �а�ɫ�������� | ��ѧ����ʽ��______ |
| �۹��� | �˳����� | �õ�������Һ |
| �������Թֱܷ�ȡ������������Һ��ԭӲˮ��Ʒ���ֱ�μ�______���� | ʢ����Һ���Թ���______ | ֤��Ӳˮ������ |
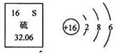 ��2010?�����ͼ��Ԫ�����ڱ��е���Ԫ�ؼ���ԭ�ӽṹʾ��ͼ������˵����ȷ���ǣ�������
��2010?�����ͼ��Ԫ�����ڱ��е���Ԫ�ؼ���ԭ�ӽṹʾ��ͼ������˵����ȷ���ǣ�������