��Ŀ����
������У����ڵ������ǡ�������̼������н��ܼ��š�������ش�
��1�������������ж�����̼��������������ԭ����________����Ȼ�������Ķ�����̼����Ҫ;����________��
��2����ѧ�������о���������̼ת��Ϊ���顢�״���CH3OH�������ᣨHCOOH���Ȼ���ԭ�ϣ���Щ����ԭ������________���������л������
��3�������������ϡ���̼���á��������________��
A���������̭�ߺ��ܡ�����Ⱦ��ҵ
B��������չ���
C���о��ͷ�����Դ�����ͳ��Դ
D���Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ�
��4����������һ�������з��ϡ�����̼�ŷš�������________��
��5������ʯ�Ͳ�Ʒ�����У�ͨ������������࣬ij������������15%������������Һϴ�����ᣬ����������������Һ40g��ϴ�Ӻ����Һ�����ԣ���д���÷�Ӧ�Ļ�ѧ��ʽ________���������Ƶ���Է�������Ϊ________������������Һ�к����к���Ԫ��________g����Щ�������ƿ����кʹ���________��������������Ϊ10%��������ˮ��
�⣺��1���������е�֪ʶ����֪������������ʯȼ�ϵĴ���ʹ�ã����¿����ж�����̼�ĺ����������ӣ���ɫֲ��Ĺ�����������Ķ�����̼����Ҫ;���������ʯȼ�ϵĴ���ʹ�ã���ɫֲ��Ĺ�����ã�
��2�����顢�״���CH3OH�������ᣨHCOOH�����Ǻ���̼Ԫ�ص����ʣ������л������л��
��3��A���������̭�ߺ��ܡ�����Ⱦ��ҵ�ܼ��ٶ�����̼���ŷţ���A��ȷ��
B��������չ��磬������ʯȼ�ϵ�ʹ�ã����ӿ����ж�����̼�ĺ�������B����
C���о��ͷ�����Դ�����ͳ��Դ���ܼ��ٶ�����̼���ŷţ���C��ȷ��
D���Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ磬�ܼ��ٶ�����̼���ŷţ���D��ȷ��
���ACD��
��4�����Ŷ�������������ܼ��ٻ�ʯȼ�ϵ�ʹ�ã��Ӷ����ٶ�����̼���ŷţ�������ž�������������
��5�����������������ᷴӦ���������ƺ�ˮ���������Ƶ���Է�������Ϊ��23+16+1=40��
Ҫ������������Һ����Ԫ�ص�������������Ҫ�����Һ���������Ƶ�������Ȼ�����������������Ԫ�ص��������������40g��15%��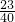 =3.45g��
=3.45g��
����Դ���������������Ϊ10%��������ˮ������Ϊx
2NaOH+H2SO4�TNa2SO4+2H2O
80 98
40g��15% 10%��x
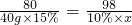
x=73.5g
���2NaOH+H2SO4=Na2SO4+2H2O��40��3.45��73.5��
�������������ʵ������Լ����ʼ�ķ�Ӧ���з�������ʯȼ�ϵĴ���ʹ�ã����¿����ж�����̼�ĺ������ߣ���Ȼ�������Ķ�����̼����Ҫ;������ɫֲ��Ĺ�����ã��л�����ָ����̼Ԫ�ص����ʣ���̼������Ǽ��ٿ����ж�����̼�ĺ�������������������ϡ�����㷴Ӧ�Ļ�ѧ����ʽ��������غ㶨�ɼ��ɼ�����й����ʵ�����
���������⿼���˻�ѧ�뻷��������֪ʶ����ɴ��⣬�����������ʵ������Լ������غ㶨�ɽ��У�
��2�����顢�״���CH3OH�������ᣨHCOOH�����Ǻ���̼Ԫ�ص����ʣ������л������л��
��3��A���������̭�ߺ��ܡ�����Ⱦ��ҵ�ܼ��ٶ�����̼���ŷţ���A��ȷ��
B��������չ��磬������ʯȼ�ϵ�ʹ�ã����ӿ����ж�����̼�ĺ�������B����
C���о��ͷ�����Դ�����ͳ��Դ���ܼ��ٶ�����̼���ŷţ���C��ȷ��
D���Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ磬�ܼ��ٶ�����̼���ŷţ���D��ȷ��
���ACD��
��4�����Ŷ�������������ܼ��ٻ�ʯȼ�ϵ�ʹ�ã��Ӷ����ٶ�����̼���ŷţ�������ž�������������
��5�����������������ᷴӦ���������ƺ�ˮ���������Ƶ���Է�������Ϊ��23+16+1=40��
Ҫ������������Һ����Ԫ�ص�������������Ҫ�����Һ���������Ƶ�������Ȼ�����������������Ԫ�ص��������������40g��15%��
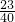 =3.45g��
=3.45g������Դ���������������Ϊ10%��������ˮ������Ϊx
2NaOH+H2SO4�TNa2SO4+2H2O
80 98
40g��15% 10%��x
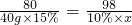
x=73.5g
���2NaOH+H2SO4=Na2SO4+2H2O��40��3.45��73.5��
�������������ʵ������Լ����ʼ�ķ�Ӧ���з�������ʯȼ�ϵĴ���ʹ�ã����¿����ж�����̼�ĺ������ߣ���Ȼ�������Ķ�����̼����Ҫ;������ɫֲ��Ĺ�����ã��л�����ָ����̼Ԫ�ص����ʣ���̼������Ǽ��ٿ����ж�����̼�ĺ�������������������ϡ�����㷴Ӧ�Ļ�ѧ����ʽ��������غ㶨�ɼ��ɼ�����й����ʵ�����
���������⿼���˻�ѧ�뻷��������֪ʶ����ɴ��⣬�����������ʵ������Լ������غ㶨�ɽ��У�

��ϰ��ϵ�д�
�����Ŀ