��Ŀ����
��������߶ȹ�ע���ӽ���̽��������������Դ--��ȼ�����״����뵽�ҹ���Դ�滮֮�У��ݱ�������ѧ�����������һ���������Ĺ�̬���ʣ��������ʵijɷ��Ǽ���ˮ���CH4?xH2O������Ϊ��ȼ����ͨ������£��������ʲ��ȶ����ֽ⡢��ȼ���ڵ��»��ѹ�³ʹ�̬���䴢������ú��ʯ�͵��ܴ������Ǻ���ǰ;�Ĵ�������Դ��
��1�����������ȼ����Ϊ��Դ��ʲô�ŵ㣿______��
��2����д������ˮ�����е���Ҫ�ɷּ����ڿ�����ȼ�յķ���ʽ______��
��3����ѧ�Ҷ����ҹ��Ϻ������ֵĿ�ȼ������ֽ����о�����京̼������������Ϊ7.5%����x��ֵΪ______��
��4��������־���ĽṹΪƽ��ÿ46��ˮ���ӽ�8������ÿ��������һ��������ӻ�һ�������ˮ���ӣ��������־����й���______�ַ��ӣ���ÿ8��������6�������2��ˮ���ӣ�������8������ɵ������У�������ˮ�ķ��Ӹ�������______��
�⣺��1������ȼ������δ���ྻ������Դ���䴢������ú��ʯ�͵��ܴ�����˵���䴢�������ˮ������������ȼ�պ��������κβ�������Ⱦ��ú��ʯ�͡���Ȼ����ҪС�ö࣮
��2����Ȼ������Ҫ�ɷ��Ǽ��飨CH4�����ڿ�����ȼ�պ����ɶ�����̼��ˮ����Ӧ�Ļ�ѧ����ʽΪCH4+2O2 CO2+2H2O��
CO2+2H2O��
��3������������й�ϵʽ��7.5%=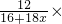 100%��x=8��
100%��x=8��
��4���������֪�����־����й���������Ӻ�ˮ�������ַ��ӣ�������ƽ��ÿ46��H2O������8������8������6��������CH4���ӣ�����2�����������H2O������䣬��ˣ���������48��ˮ���ӣ�����CH4���ӣ�����6CH4?48H2O��CH4?8H2O��������ˮ�ķ��Ӹ�������1��8��
�ʴ�Ϊ����1����������ࣻ��2��CH4+2O2 CO2+2H2O����3��8����4��2��1��8��
CO2+2H2O����3��8����4��2��1��8��
��������1�������漰������Դ���γ�ԭ���̽���Լ����ŵ��̽�����⣬����Ŀ������Ϣ�����ƶϼ��ɣ�
��2�������ⷴӦ��Ϊ������������������Ϊ������̼��ˮ����Ӧ����Ϊ��ȼ���ʿ���д��ѧ����ʽ��
��3������̼Ԫ�ص������������й�ϵʽ��⼴�ɣ�
��4���������⣬�ڡ���ȼ���������У�ÿ8������46��H2O���ӹ��ɿ�ܣ���������6��CH4���Ӻ�2��H2O���ӣ����ж�ÿ8������48��H2O���Ӻ�6��CH4���ӣ��ݴ��ƶϿɱ�ʾ��ȼ����ƽ����ɵĻ�ѧʽ��
����������һ��Ӧ������Ϣ�⣬�����Ķ����ϴ���Ҫ��������Ϣ�ܶ࣬����ѧ�����ܲ���������Ϣ������������������ʱҪ���������Ϣ��ʵ�ʣ�����¾�֪ʶ����Ҫ����������з��������
��2����Ȼ������Ҫ�ɷ��Ǽ��飨CH4�����ڿ�����ȼ�պ����ɶ�����̼��ˮ����Ӧ�Ļ�ѧ����ʽΪCH4+2O2
 CO2+2H2O��
CO2+2H2O����3������������й�ϵʽ��7.5%=
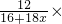 100%��x=8��
100%��x=8����4���������֪�����־����й���������Ӻ�ˮ�������ַ��ӣ�������ƽ��ÿ46��H2O������8������8������6��������CH4���ӣ�����2�����������H2O������䣬��ˣ���������48��ˮ���ӣ�����CH4���ӣ�����6CH4?48H2O��CH4?8H2O��������ˮ�ķ��Ӹ�������1��8��
�ʴ�Ϊ����1����������ࣻ��2��CH4+2O2
 CO2+2H2O����3��8����4��2��1��8��
CO2+2H2O����3��8����4��2��1��8����������1�������漰������Դ���γ�ԭ���̽���Լ����ŵ��̽�����⣬����Ŀ������Ϣ�����ƶϼ��ɣ�
��2�������ⷴӦ��Ϊ������������������Ϊ������̼��ˮ����Ӧ����Ϊ��ȼ���ʿ���д��ѧ����ʽ��
��3������̼Ԫ�ص������������й�ϵʽ��⼴�ɣ�
��4���������⣬�ڡ���ȼ���������У�ÿ8������46��H2O���ӹ��ɿ�ܣ���������6��CH4���Ӻ�2��H2O���ӣ����ж�ÿ8������48��H2O���Ӻ�6��CH4���ӣ��ݴ��ƶϿɱ�ʾ��ȼ����ƽ����ɵĻ�ѧʽ��
����������һ��Ӧ������Ϣ�⣬�����Ķ����ϴ���Ҫ��������Ϣ�ܶ࣬����ѧ�����ܲ���������Ϣ������������������ʱҪ���������Ϣ��ʵ�ʣ�����¾�֪ʶ����Ҫ����������з��������

��ϰ��ϵ�д�
�����Ŀ
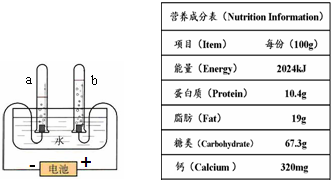 �������£����ס��͡��Ρ������ס��裮��ʾ�˼�ͥ��Ҫ�ĸ��ֱ���Ʒ���ݹۻ�ѧѧ�Ƶ�ѧϰ�������������ʵ���ϵҲ�൱���У����Ծټ�����
�������£����ס��͡��Ρ������ס��裮��ʾ�˼�ͥ��Ҫ�ĸ��ֱ���Ʒ���ݹۻ�ѧѧ�Ƶ�ѧϰ�������������ʵ���ϵҲ�൱���У����Ծټ�����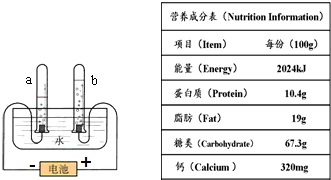 �������£����ס��͡��Ρ������ס��裮��ʾ�˼�ͥ��Ҫ�ĸ��ֱ���Ʒ���ݹۻ�ѧѧ�Ƶ�ѧϰ�������������ʵ���ϵҲ�൱���У����Ծټ�����
�������£����ס��͡��Ρ������ס��裮��ʾ�˼�ͥ��Ҫ�ĸ��ֱ���Ʒ���ݹۻ�ѧѧ�Ƶ�ѧϰ�������������ʵ���ϵҲ�൱���У����Ծټ�����