��Ŀ����
����������Ƽ��������ȶ��벻����ѧ��
��ʳƷ��ҩ�ﰲȫһֱ�����ǹ�ע�Ļ��⣮
��1��2012��11��17�վ���������Ƶ���ع���ù�ҵ�����Ƴɵ�ҽ�á������ҡ���ȫ������Ϥ�����л��е��ؽ������������к�������ĸ���ָ
��2������ʢ���ĺ���Ʒ��Ϻ�и�������Ӫ���أ����ࡢ��֬�������ʣ��е�
���ϵ�Ӧ���뷢չ����������ǵ���������������ֲ��ϵĶ�Ӧ��ĸ��գ�
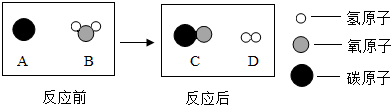
A���л��ϳɲ��� B���������� C����Ȼ�л��߷��Ӳ���
��1��������Ʒ����
��2�����������
��3����������������ǡ�����ɻ��ȵ��ѺϽ�����
���ܡ����š���̼�������ǵ�������������ɣ�
��1������
��2��úȼ��ʱ�����ʵ������ʣ����Լ���
���������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ����������ˮ����������Ŀǰ���õ�Һ�����⣬�������Ȱ���NH2Cl����������O3����
��1��O3����Ԫ�صĻ��ϼ���
��2��NH2Cl��
��3��NH2Cl������ʱ��������ӦNH2Cl+X�TNH3+HClO������X�Ļ�ѧʽ��
��ʳƷ��ҩ�ﰲȫһֱ�����ǹ�ע�Ļ��⣮
��1��2012��11��17�վ���������Ƶ���ع���ù�ҵ�����Ƴɵ�ҽ�á������ҡ���ȫ������Ϥ�����л��е��ؽ������������к�������ĸ���ָ
Ԫ��
Ԫ��
���Ԫ�ء����ʡ���Ԫ�ء�������2������ʢ���ĺ���Ʒ��Ϻ�и�������Ӫ���أ����ࡢ��֬�������ʣ��е�
������
������
������Ʒ�����ñ��ʣ�����
����
����ܡ����ܡ����ü�ȩ��Һ���ݺ���Ʒ�������ʣ����ϵ�Ӧ���뷢չ����������ǵ���������������ֲ��ϵĶ�Ӧ��ĸ��գ�
A���л��ϳɲ��� B���������� C����Ȼ�л��߷��Ӳ���
��1��������Ʒ����
A
A
����2�����������
C
C
����3����������������ǡ�����ɻ��ȵ��ѺϽ�����
B
B
�����ܡ����š���̼�������ǵ�������������ɣ�
��1������
̫����
̫����
����һ��������Դ���������Բ��ֽ����ʯ��Դ��ȱ���⣬�����Լ��ٶԻ�������Ⱦ����2��úȼ��ʱ�����ʵ������ʣ����Լ���
SO2
SO2
��д��ѧʽ������͵�����������к������ŷš���Ч����������γɣ����������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ����������ˮ����������Ŀǰ���õ�Һ�����⣬�������Ȱ���NH2Cl����������O3����
��1��O3����Ԫ�صĻ��ϼ���
O
O
����2��NH2Cl��
3
3
�������֣���Ԫ����ɣ�������Ԫ������Ԫ�ص���������71��4
71��4
����3��NH2Cl������ʱ��������ӦNH2Cl+X�TNH3+HClO������X�Ļ�ѧʽ��
H2O
H2O
���������������е�֪ʶ���з����������к��е���Ԫ�أ���Ϻ�и��������ʣ���ȩ�ж����������ڽ���ˮ��Ʒ�����ݲ��ϵ����������𣻿ɹ��������õ�����Դ��̫���ܡ����ܡ���ϫ�ܡ������ܡ����ܵȵȣ�ú�к�����Ԫ�أ�ȼ�������ɶ�������
������Ԫ�صĻ��ϼ�Ϊ��ۣ��ص�������Ϊ���ԭ����������ԭ�Ӹ���֮�ȣ��������غ㶨�ɿ�֪����Ӧǰ��Ԫ������䣬ԭ�Ӹ������䣮���������ݿ��Ƴ�X�Ļ�ѧʽ��
������Ԫ�صĻ��ϼ�Ϊ��ۣ��ص�������Ϊ���ԭ����������ԭ�Ӹ���֮�ȣ��������غ㶨�ɿ�֪����Ӧǰ��Ԫ������䣬ԭ�Ӹ������䣮���������ݿ��Ƴ�X�Ļ�ѧʽ��
����⣺I����1�������к��еĸ�ָ���Ǹ�Ԫ�أ����Ԫ�أ�
��2����Ϻ�и��������ʣ���ȩ�ж����������ڽ���ˮ��Ʒ����������ʣ����ܣ�
��1�����������л��ϳɲ��ϣ����A��
��2��������ë������Ȼ�л��߷��Ӳ��ϣ����C��
��3���ѺϽ����ڽ������ϣ����B��
��1���ɹ��������õ�����Դ��̫���ܡ����ܡ���ϫ�ܡ������ܡ����ܵȵȣ����̫���ܣ�����һ��ɣ�����2��ú�к�����Ԫ�أ�ȼ�������ɶ���������Ⱦ���������SO2
���������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ����������ˮ����������Ŀǰ���õ�Һ�����⣬�������Ȱ���NH2Cl��������O3
��1��������Ԫ�صĻ��ϼ�Ϊ��ۣ�O3Ϊ����Ԫ�ػ��ϼ�Ϊ�㣻�ʴ�Ϊ��0
��2��NH2Cl��������Ԫ�أ�������Ԫ������Ԫ�ص�������Ϊ��35.5����1��2��=71��4����Ϊ��71��4
��3������������ѧ����ʽ���Կ�������ѧ����ʽ���ұ߱���߶����2����ԭ�Ӻ�1����ԭ�ӣ����������غ㶨���е�Ԫ��������ԭ�Ӹ���������ص㣬��֪�����ԭ��ȫ������1��X�����У���X�Ļ�ѧʽΪH2O���ʴ�Ϊ��H2O
�ʴ�Ϊ����1��Ԫ�أ�2�������ʣ����ܣ�
��1��A����2��C����3��B��
��1��̫���ܣ���2��SO2
������1��O����2��3��71��4����3��H2O
��2����Ϻ�и��������ʣ���ȩ�ж����������ڽ���ˮ��Ʒ����������ʣ����ܣ�
��1�����������л��ϳɲ��ϣ����A��
��2��������ë������Ȼ�л��߷��Ӳ��ϣ����C��
��3���ѺϽ����ڽ������ϣ����B��
��1���ɹ��������õ�����Դ��̫���ܡ����ܡ���ϫ�ܡ������ܡ����ܵȵȣ����̫���ܣ�����һ��ɣ�����2��ú�к�����Ԫ�أ�ȼ�������ɶ���������Ⱦ���������SO2
���������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ����������ˮ����������Ŀǰ���õ�Һ�����⣬�������Ȱ���NH2Cl��������O3
��1��������Ԫ�صĻ��ϼ�Ϊ��ۣ�O3Ϊ����Ԫ�ػ��ϼ�Ϊ�㣻�ʴ�Ϊ��0
��2��NH2Cl��������Ԫ�أ�������Ԫ������Ԫ�ص�������Ϊ��35.5����1��2��=71��4����Ϊ��71��4
��3������������ѧ����ʽ���Կ�������ѧ����ʽ���ұ߱���߶����2����ԭ�Ӻ�1����ԭ�ӣ����������غ㶨���е�Ԫ��������ԭ�Ӹ���������ص㣬��֪�����ԭ��ȫ������1��X�����У���X�Ļ�ѧʽΪH2O���ʴ�Ϊ��H2O
�ʴ�Ϊ����1��Ԫ�أ�2�������ʣ����ܣ�
��1��A����2��C����3��B��
��1��̫���ܣ���2��SO2
������1��O����2��3��71��4����3��H2O
���������⿼���˻�ѧ�������֪ʶ����ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
�����Ŀ