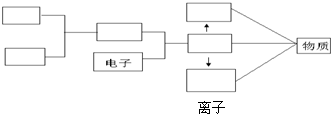
��Ŀ����
�������д����������ʹ��������Σ���ԣ�
��1����ȼ�ŵľƾ��Ƶ�ȼ��һյ�ƾ��� ��
��2����ʢ��Һ���������
���Թܼ��� ��
��3������ʯ���������ձ�������Ȧ��ֱ�Ӽ��� ��
��4������ڸ�ˮ���Թ�ʢ��ʽ̼��ͭ������� ��
��5���ι�ȡ���Լ���ƽ�Ż��������� ��
��6��ȡϸ��ƿ���ҩҺʱ����ǩδ�������� ��
��7������Һ��ʱ���Թܿڶ������˵ķ��� ��
��8�����Ⱥ���Թ�ֱ�ӷ���ʵ������ ��
��1����ȼ�ŵľƾ��Ƶ�ȼ��һյ�ƾ���
��2����ʢ��Һ���������
| 1 |
| 3 |
��3������ʯ���������ձ�������Ȧ��ֱ�Ӽ���
��4������ڸ�ˮ���Թ�ʢ��ʽ̼��ͭ�������
��5���ι�ȡ���Լ���ƽ�Ż���������
��6��ȡϸ��ƿ���ҩҺʱ����ǩδ��������
��7������Һ��ʱ���Թܿڶ������˵ķ���
��8�����Ⱥ���Թ�ֱ�ӷ���ʵ������
���㣺��������-�ƾ���,���ڼ��ȵ�����,Һ��ҩƷ��ȡ��,���Թ���Ĺ������,���Թ����Һ�����
ר�⣺������������ѧʵ���������
��������1���Ӿƾ����лӷ��ԣ�Ϊ��ֹʧ��ȥ�������
��2���Ӹ��Թ����Һ�����ʱ��ע������ȥ�������
��3���ӷ�ֹ�ձ������Ȳ�������ը��ȥ�������
��4���ɸ��Թ��ڵĹ��������Ҫע��������з�����
��5��ʹ��ʱ��ͷ���ϣ��ܿ����£���ֹƽ�Ż��ã�
��6�����ݱ�ǩ���������ģ�ҩҺ��������ǩ�ϣ�
��7�����ݸ��Թ��е�Һ�����ʱ��ע��������з������
��8���մӾƾ��ƻ����ϳ��µ��Թܣ���������ϴ�ӣ�Ҳ����ֱ�ӷ�������̨�ϣ�
��2���Ӹ��Թ����Һ�����ʱ��ע������ȥ�������
��3���ӷ�ֹ�ձ������Ȳ�������ը��ȥ�������
��4���ɸ��Թ��ڵĹ��������Ҫע��������з�����
��5��ʹ��ʱ��ͷ���ϣ��ܿ����£���ֹƽ�Ż��ã�
��6�����ݱ�ǩ���������ģ�ҩҺ��������ǩ�ϣ�
��7�����ݸ��Թ��е�Һ�����ʱ��ע��������з������
��8���մӾƾ��ƻ����ϳ��µ��Թܣ���������ϴ�ӣ�Ҳ����ֱ�ӷ�������̨�ϣ�
����⣺��1�����ھƾ����лӷ��ԣ�Ϊ��ֹʧ�𣬾��Խ�ֹ�þƾ�����ȼ��һ�ƾ��ƣ�
�ʴ�Ϊ������ʧ��
��2�����Թ����Һ�����ʱ����ֹ�Թ��ڵ�Һ����ڽ������ˣ��Թ����Һ�岻Ӧ�����Թ��ݻ���
��
�ʴ�Ϊ��Һ����ڽ������ˣ�
��3��Ϊ��ֹ�ձ������Ȳ�������ը�ѣ����ձ�����ʱҪ����ʯ������
�ʴ�Ϊ���ձ�ը�ѣ�
��4�����Թܼ���ʱ���Թ���ڸ���ˮ�飬��ʹ�Թ����Ȳ�������ʹ�Թ�ը�ѣ�
�ʴ�Ϊ���Թ�ը�ѣ�
��5���ι�ȡ���Լ���ƽ�Ż��ã�Һ���Լ����뽺ͷ��ʹ��ͷ�ܸ�ʴ��ͷ������ʴ�����Һ��
�ʴ�Ϊ��Һ���Լ����뽺ͷ��ʹ��ͷ�ܸ�ʴ��ͷ������ʴ�����Һ��
��6���㵹ϸ��ƿ���ҩҺʱ����ǩû�������ģ��㵹Һ��ʱ�IJ���Һ��������ǩ�ϣ���ʴ��ǩ��
�ʴ��ǣ��㵹Һ��ʱ�IJ���Һ��������ǩ�ϣ���ʴ��ǩ��
��7������Һ��ʱ���Թܿڶ������˵ķ��������Һ����ڽ���ʱ���ˣ�
�ʴ�Ϊ��Һ����ڽ���ʱ���ˣ�
��8���մӾƾ��ƻ����ϳ��µ��Թ�Ӧ����ʯ�����ϣ��Է��Թ�����ը�ѣ�
�ʴ��ǣ��Թ�ը�ѣ�
�ʴ�Ϊ������ʧ��
��2�����Թ����Һ�����ʱ����ֹ�Թ��ڵ�Һ����ڽ������ˣ��Թ����Һ�岻Ӧ�����Թ��ݻ���
| 1 |
| 3 |
�ʴ�Ϊ��Һ����ڽ������ˣ�
��3��Ϊ��ֹ�ձ������Ȳ�������ը�ѣ����ձ�����ʱҪ����ʯ������
�ʴ�Ϊ���ձ�ը�ѣ�
��4�����Թܼ���ʱ���Թ���ڸ���ˮ�飬��ʹ�Թ����Ȳ�������ʹ�Թ�ը�ѣ�
�ʴ�Ϊ���Թ�ը�ѣ�
��5���ι�ȡ���Լ���ƽ�Ż��ã�Һ���Լ����뽺ͷ��ʹ��ͷ�ܸ�ʴ��ͷ������ʴ�����Һ��
�ʴ�Ϊ��Һ���Լ����뽺ͷ��ʹ��ͷ�ܸ�ʴ��ͷ������ʴ�����Һ��
��6���㵹ϸ��ƿ���ҩҺʱ����ǩû�������ģ��㵹Һ��ʱ�IJ���Һ��������ǩ�ϣ���ʴ��ǩ��
�ʴ��ǣ��㵹Һ��ʱ�IJ���Һ��������ǩ�ϣ���ʴ��ǩ��
��7������Һ��ʱ���Թܿڶ������˵ķ��������Һ����ڽ���ʱ���ˣ�
�ʴ�Ϊ��Һ����ڽ���ʱ���ˣ�
��8���մӾƾ��ƻ����ϳ��µ��Թ�Ӧ����ʯ�����ϣ��Է��Թ�����ը�ѣ�
�ʴ��ǣ��Թ�ը�ѣ�
������������Ҫ�˽��������ע������˽�߱������Ļ�ѧʵ�鼼����ѧϰ��ѧ�ͽ��л�ѧ̽����Ļ����ͱ�֤��ֻ�����ջ�ѧʵ��Ļ������ܣ����ܰ�ȫ��ȷ�ؽ��л�ѧʵ�飮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��̨��ʱ����������������������ѹ��С��ijͬѧ�����ݵ��ݶ����Ʒ�����ͼ��ʾ���������ԭ�������ͬ���ǣ�������
A��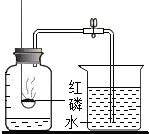 ��ͼ������ȴ����ӣ�ˮ��������ƿ |
B��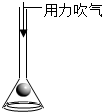 ��ͼ�����������´�������ȴ������©���� |
C��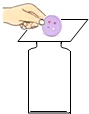 ��ͼ��������Ƭ���ٵ���ȥ������ȴ����ƿ�� |
D�� ��ͼ�����������ȼ���糵�ͻ�ת���� |
�����и����Ϻ�����ˮ���õ���ɫ��Һ���ǣ�������
| A��MgSO4��KOH��Na2SO4 |
| B��FeCl3��NaOH��NaCl |
| C��Na2SO4��NaCl��NaOH |
| D��BaCl2��Na2SO4��NaCl |
��������������һ���Ż�����������족�����������ɷ������ֱ���ϡ���ᡢ�Ȼ�����Һ��ʯ��ˮ����ͼ�������ˡ�����֡����ϼ��ɳ��������������족�ֱ�ȥ���ʡ����ǡ������ѡ����ǡ���������������������ġ����족�ǣ�������
A�� ��ɫʯ����Һ�����족 |
B�� ϡ���ᡰ���족 |
C�� ��ɫ��̪��Һ�����족 |
D�� ������Һ�����족 |