��Ŀ����
��2013?����ģ�⣩Сޱͬѧͨ���������ϵ�֪����������һ����ɫ���壬������ˮ�������л��ᣬ�������ͨ�ԣ�����ͼ���䷽����һƿ�����������ˮ������ͼ����Ϣ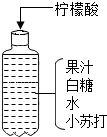
��1���û�ѧ����ش𣺢�����������ˮ���ܽ����
��2��Сޱͬѧ��������������ˮ����һ�ྻ���Թ��У�Ȼ��μ���ɫʯ����Һ������ʯ����Һ���ɫ���ݴˣ�Сޱ�϶�������ȷʵ�������ԣ�����Ϊ��ʵ������Ƿ����ܣ�����

��1���û�ѧ����ش𣺢�����������ˮ���ܽ����
H+
H+
�����Ծ������ͨ�ԣ����ǵ���Ҫ�ɷ�C12H22O11�����л���������ᶼ����H
H
Ԫ�أ�����ˮƿ�Dz����������Ҫ�ɷ���CO2
CO2
������������������NaHCO3
NaHCO3
��Ӧ���ɣ���2��Сޱͬѧ��������������ˮ����һ�ྻ���Թ��У�Ȼ��μ���ɫʯ����Һ������ʯ����Һ���ɫ���ݴˣ�Сޱ�϶�������ȷʵ�������ԣ�����Ϊ��ʵ������Ƿ����ܣ�����
��ˮ�е�̼��Ҳ��ʹʯ����ɫ
��ˮ�е�̼��Ҳ��ʹʯ����ɫ
����������1���������ͨ�Ե�ԭ���Լ�����̼���εķ�Ӧ���ɷ������
��2�����ݶ�����̼����ˮ������̼�ᣬ��̼����ʹʯ����������
��2�����ݶ�����̼����ˮ������̼�ᣬ��̼����ʹʯ����������
����⣺��1����������������ˮ���ܽ����H+�����Ծ������ͨ�ԣ�C12H22O11�����л���������ᶼ��HԪ�أ�����ˮƿ�Dz����������Ҫ�ɷ��Ƕ�����̼������������������С�մ�NaHCO3����Ӧ�Ƶã�
��2��������ˮ�к��ж�����̼��������̼�ܹ���ˮ��Ӧ����̼�ᣬ̼��Ҳ��ʹʯ���죬���Ը��ƶϲ������ܣ�
�ʴ�Ϊ����1��H+��H��CO2��NaHCO3����2����ˮ�е�̼��Ҳ��ʹʯ����ɫ��
��2��������ˮ�к��ж�����̼��������̼�ܹ���ˮ��Ӧ����̼�ᣬ̼��Ҳ��ʹʯ���죬���Ը��ƶϲ������ܣ�
�ʴ�Ϊ����1��H+��H��CO2��NaHCO3����2����ˮ�е�̼��Ҳ��ʹʯ����ɫ��
�����������ѶȲ��Ǻܴ���Ҫ����������йصĻ�ѧ���ʣ�����ѧ�����й�֪ʶ��Ӧ��������

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ����������ϰ��ϵ�д�
�Ƹ�С״Ԫ����������ϰ��ϵ�д� �ɹ�ѵ���ƻ�ϵ�д�
�ɹ�ѵ���ƻ�ϵ�д� ����ѵ����ֱͨ�п�����ϵ�д�
����ѵ����ֱͨ�п�����ϵ�д� һ���㶨ϵ�д�
һ���㶨ϵ�д� ��У��ҵ��ϵ�д�
��У��ҵ��ϵ�д�
�����Ŀ