��Ŀ����
2013��3��22���ǵڶ�ʮһ�조����ˮ�ա������Ϲ�ȷ��2013�ꡰ����ˮ�ա������������ǡ�ˮ��������ˮ������֮Դ������Ӧ���˽�ˮ������ˮ��Դ��
��1������ɽǶȿ���ˮ���� ��ɵġ�
��2���ӽṹ�Ƕȿ���ˮ���� ���ɵġ�
 ��3���ӱ仯�Ƕȿ����仯Ī���ˮ�����Ŵ���Ȼ�����档 ��ˮ�μ�ֲ�������õĻ�ѧ����ʽΪ��6CO2+6H2O 6O2+X����X�Ļ�ѧʽ
��3���ӱ仯�Ƕȿ����仯Ī���ˮ�����Ŵ���Ȼ�����档 ��ˮ�μ�ֲ�������õĻ�ѧ����ʽΪ��6CO2+6H2O 6O2+X����X�Ļ�ѧʽ
��ˮͨ��ֽ�����δ�����������Դ——�������ǽ� ��ת��Ϊ��ѧ�ܣ����÷��������ܣ���ҪѰ�Ҹ�Ϊ��Ч�Ľ��ܷ����ٽ�ˮ�ķֽ⣻��ͼ1��ˮ�ĵ��װ�ã����������Դ �������IJ������ڷų���
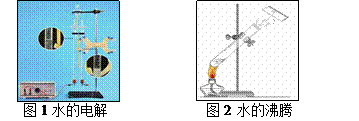
����ͼ1��ͼ2��ʾˮ���������ֱ仯�У����۽Ƕȿ������ֱ仯�ı����������� ����4������Դ�Ƕȿ����������Լ��7l����ˮ���ǣ����ɹ��������õĵ�ˮ����ȴ����1������
��4����ˮ��Դ��ÿ����������κ�����������Ϊ���ڽ�Լ��ˮ���� ������ţ���
A��ϴ�ֲ�ϴ��Һʱ������ˮ��ͷ B�����ֹر�ˮ��ͷ
C��������ˮ����Ϊ���ϳ�ˮ���� D����ϴ��ˮ�����
��5������;�Ƕȿ���ˮ�����ԭ����Ҫ�� ��ˮ�����ܼ��������е�ζƷ����ˮ�У������γ���Һ���� ������ţ���
A��ʳ�� B������ C��ζ�� D������
��1����Ԫ�غ���Ԫ�أ���2��ˮ���ӣ���3����C6H12O6���ڵ硢�����۱仯������Ƿ����ı䣻��4��BD����5�����Ϳ�ȼ����¶ȵ��Ż�����¡�D��

 ����5��2���ϵ�д�
����5��2���ϵ�д� ��2013?��ˮ��һģ��2013��3��22���ǵڶ�ʮһ�조����ˮ�ա��� ����������ǡ�ˮ��������ˮ����������������������أ������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ���±����ҹ��䲼����������ˮˮ�ʱ��IJ������ݣ�
��2013?��ˮ��һģ��2013��3��22���ǵڶ�ʮһ�조����ˮ�ա��� ����������ǡ�ˮ��������ˮ����������������������أ������°桶��������ˮ���������ӽ���7��1����ǿ��ʵʩ���±����ҹ��䲼����������ˮˮ�ʱ��IJ������ݣ�
 ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� 2013��3��22���ǵ�21�조����ˮ�ա����������������ǡ�ˮ��������Water Cooperation�������������ѧ���ش��������⣮
2013��3��22���ǵ�21�조����ˮ�ա����������������ǡ�ˮ��������Water Cooperation�������������ѧ���ش��������⣮