��Ŀ����
��1���������ˡ����ơ��������л��ȹܣ�����������ͭ������ȼ��Ͳ��࣬ʹ������µ������ܹ�����������Ҫ�������˽���ͭ�� �ԣ�
��2������������ȼ������Ȼ������ȼ�յĻ�ѧ����ʽΪ ��
��3���������˻᳡�ݡ��������ṹΪ�ֽṹ��Ϊ��ֹ�������⣬�ɲ�ȡ�ķ���֮һ�� ����ҵ��ұ������ԭ��֮һ����һ����̼��ԭ���������÷�Ӧ�Ļ�ѧ����ʽΪ ��
��2������������ȼ������Ȼ������ȼ�յĻ�ѧ����ʽΪ
��3���������˻᳡�ݡ��������ṹΪ�ֽṹ��Ϊ��ֹ�������⣬�ɲ�ȡ�ķ���֮һ��
���㣺�������������ʼ���;,һ����̼��ԭ������,������ʴ�������������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,����ȼ�ϵ�ʹ������Ի�����Ӱ��
ר�⣺�������������
��������1��ͭ���д����ԣ�
��2����Ȼ������Ҫ�ɷ��Ǽ��飬д������ȼ�յĻ�ѧ����ʽ��
��3�����ݷ�ֹ������ķ�����������ԭ�������ش�
��2����Ȼ������Ҫ�ɷ��Ǽ��飬д������ȼ�յĻ�ѧ����ʽ��
��3�����ݷ�ֹ������ķ�����������ԭ�������ش�
����⣺��1������ͭ���д����ԣ��������ˡ����ơ��������еĻ��ȹܣ�����������ͭ�Ĵ����ԣ�
��2������������ȼ������Ȼ������Ȼ������Ҫ�ɷ��Ǽ��飬����ȼ�������˶�����̼��ˮ��ȼ�յĻ�ѧ����ʽΪ��CH4+2O2
CO2+2H2O��
��3��Ϊ��ֹ�������⣬�ɲ�ȡ�ķ���֮һ��ˢ���Ϳ�͵ȣ���ҵ��ұ������ԭ��֮һ����һ����̼��ԭ���������÷�Ӧ�Ļ�ѧ����ʽΪ��Fe2O3+3CO
2Fe+3CO2��
�ʴ�Ϊ����1�������ԣ���2��ˢ�ᣬCH4+2O2
CO2+2H2O����3��Fe2O3+3CO
2Fe+3CO2��
��2������������ȼ������Ȼ������Ȼ������Ҫ�ɷ��Ǽ��飬����ȼ�������˶�����̼��ˮ��ȼ�յĻ�ѧ����ʽΪ��CH4+2O2
| ||
��3��Ϊ��ֹ�������⣬�ɲ�ȡ�ķ���֮һ��ˢ���Ϳ�͵ȣ���ҵ��ұ������ԭ��֮һ����һ����̼��ԭ���������÷�Ӧ�Ļ�ѧ����ʽΪ��Fe2O3+3CO
| ||
�ʴ�Ϊ����1�������ԣ���2��ˢ�ᣬCH4+2O2
| ||
| ||
�����������ԡ����ơ����Ϊ����㣬�����˽��������ʡ�����ʴ�Ĵ�ʩ����ѧ����ʽ����д��֪ʶ���ѶȲ�������ѧ���йص�֪ʶ�������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 ��ͼ�ǡ��Ͻ��̻𡱵�������ȫ��־�����г�����ú�� �ڼ���վ����۳� ��ú��վ��Ӧ���˱�־���У�������
��ͼ�ǡ��Ͻ��̻𡱵�������ȫ��־�����г�����ú�� �ڼ���վ����۳� ��ú��վ��Ӧ���˱�־���У�������| A��ֻ�Т٢� | B��ֻ�Тڢ� |
| C��ֻ�Т٢ڢ� | D���٢ڢۢ� |
 ʵ���Ұ�ͼ��ʾװ����ȡCO2����ʵ��C02�����ʣ��Իش��������⣺
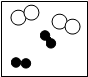
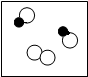
ʵ���Ұ�ͼ��ʾװ����ȡCO2����ʵ��C02�����ʣ��Իش��������⣺ ������o���ֱ��ʾ��ͬ��ԭ�ӣ����б�ʾ��������ǣ�������
������o���ֱ��ʾ��ͬ��ԭ�ӣ����б�ʾ��������ǣ�������