��Ŀ����
��2011��ɽ��Ϋ����22�⣩�����ܽ���ѧϰ��ѧ����Ҫ��������ش��������⣺
��1����һ̼��ѧ������һ��������ֻ��һ��̼ԭ�ӵĻ�����Ϊԭ�Ϻϳ�һϵ�л���ԭ�Ϻ�ȼ�ϵĻ�ѧ��
�ټ���Ļ�ѧʽ��
������ʱ��һ����̼��ԭ�������Ļ�ѧ����ʽΪ
���ڴ������ڵ������£���CO��H2�ϳɼ״���CH4O����д����Ӧ�Ļ�ѧ����ʽΪ
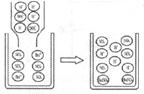
��2�������֮�������Ӧ��������֮�������������ɣ���Ӧ���ܷ�������д��������Ļ�ѧʽ ��������֮���ܷ�����Ӧ��������ͼ��д����Ӧ�Ļ�ѧ����ʽ
��
��3�������˳������Ҫ��Ӧ�á�
�ٽ�һ��пƬ���뵽CuSO4��Һ�У��۲쵽���Ե�����Ӧ�Ļ�ѧ����ʽ
��
�ڽ����ֱ���ˮ��ˮ�����ķ�Ӧ��������±���
| ���� | þ����ˮ | ������ˮ | ����ˮ���� | ͭ����ˮ | ͭ��ˮ���� |
| ��Ӧ��� | ������Ӧ | ����Ӧ | �ܷ�Ӧ | ��Ӧ | ����Ӧ |
���ݽ������˳��ͱ��е���Ϣ���ƶϡ�þ��ˮ�������ķ�Ӧ�������� ����д���ţ�
A������Ӧ B�� ������Ӧ C ����þ����ˮ��Ӧ�� D ��������ˮ������Ӧ��
����֪�ڼ��ȵ�����������ˮ������Ӧ�����������������������÷�Ӧ�Ļ�ѧ����ʽΪ
��
(1) ��CH4
��3CO+Fe2O3 2Fe+3CO2
2Fe+3CO2
(2)NH3�� Ba(NO3)2+H2SO4==BaSO4��+2HNO3
(3)��Zn+CuSO4==Cu+ZnSO4����C��D
3Fe+4H2O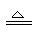 Fe3O4+4H2��
Fe3O4+4H2��
����

��2011��ɽ��Ϋ����26�⣩������������ʹ����������������������һ����Ҫ�ı�־��
��1��ÿ�������ʴ������ɾ����ʧ������Ʒ��ʴ����Ҫԭ����
��
��2��У������ȤС���ͬѧ��ȥΫ��ij������������Ʒ����ʵ�飺���ķݲ�ͬ������������Ʒ���ٶ�����ֻ���������͵���̼���ֱ�ӵ�100g����������ͬ��ϡ�����У���ַ�Ӧ��õ�ʵ���������±�����֪���ڱ�״���£�22.4LH2������Ϊ2g��
| ʵ����� | 1 | 2 | 3 | 4 |
| ������Ʒ������ / g | 2.88 | 5.76 | 9.24 | 10.28 |
| ����H2���������״���£�/L | 1.12 | 2.24 | 3.36 | m |
ͨ������ش��������⣺
������������m��ֵΪ ��
�ڸ��ݱ������ݼ���ϡ������H2SO4������������
��2011��ɽ��Ϋ����22�⣩�����ܽ���ѧϰ��ѧ����Ҫ��������ش��������⣺
��1����һ̼��ѧ������һ��������ֻ��һ��̼ԭ�ӵĻ�����Ϊԭ�Ϻϳ�һϵ�л���ԭ�Ϻ�ȼ�ϵĻ�ѧ��
�ټ���Ļ�ѧʽ��
������ʱ��һ����̼��ԭ�������Ļ�ѧ����ʽΪ
���ڴ������ڵ������£���CO��H2�ϳɼ״���CH4O����д����Ӧ�Ļ�ѧ����ʽΪ
��2�������֮�������Ӧ��������֮�������������ɣ���Ӧ���ܷ�������д��������Ļ�ѧʽ ��������֮���ܷ�����Ӧ��������ͼ��д����Ӧ�Ļ�ѧ����ʽ
��
��3�������˳������Ҫ��Ӧ�á�
�ٽ�һ��пƬ���뵽CuSO4��Һ�У��۲쵽���Ե�����Ӧ�Ļ�ѧ����ʽ
��
�ڽ����ֱ���ˮ��ˮ�����ķ�Ӧ��������±���
| ���� | þ����ˮ | ������ˮ | ����ˮ���� | ͭ����ˮ | ͭ��ˮ���� |
| ��Ӧ��� | ������Ӧ | ����Ӧ | �ܷ�Ӧ | ��Ӧ | ����Ӧ |
���ݽ������˳��ͱ��е���Ϣ���ƶϡ�þ��ˮ�������ķ�Ӧ�������� ����д���ţ�
A������Ӧ B�� ������Ӧ C ����þ����ˮ��Ӧ�� D ��������ˮ������Ӧ��
����֪�ڼ��ȵ�����������ˮ������Ӧ�����������������������÷�Ӧ�Ļ�ѧ����ʽΪ
��
��1��ÿ�������ʴ������ɾ����ʧ������Ʒ��ʴ����Ҫԭ����
��
��2�� У������ȤС���ͬѧ��ȥΫ��ij������������Ʒ����ʵ�飺���ķݲ�ͬ��������
����Ʒ���ٶ�����ֻ���������͵���̼���ֱ�ӵ�100g����������ͬ��ϡ�����У���ַ�Ӧ��õ�ʵ���������±�����֪���ڱ�״���£�22.4LH2������Ϊ2g��
| ʵ����� | 1 | 2 | 3 | 4 |
| ������Ʒ������ / g | 2.88 | 5.76 | 9.24 | 10.28 |
| ����H2���������״���£�/L | 1.12 | 2.24 | 3.36 | m |
ͨ������ش��������⣺
������������m��ֵΪ ��
�ڸ��ݱ������ݼ���ϡ������H2SO4������������
��2011��ɽ��Ϋ����26�⣩������������ʹ����������������������һ����Ҫ�ı�־��
��1��ÿ�������ʴ������ɾ����ʧ������Ʒ��ʴ����Ҫԭ����
��
��2�� У������ȤС���ͬѧ��ȥΫ��ij������������Ʒ����ʵ�飺���ķݲ�ͬ��������
����Ʒ���ٶ�����ֻ���������͵���̼���ֱ�ӵ�100g����������ͬ��ϡ�����У���ַ�Ӧ��õ�ʵ���������±�����֪���ڱ�״���£�22.4LH2������Ϊ2g��
|
ʵ����� |
1 |
2 |
3 |
4 |
|
������Ʒ������ / g |
2.88 |
5.76 |
9.24 |
10.28 |
|
����H2���������״���£�/L |
1.12 |
2.24 |
3.36 |
m |
ͨ������ش��������⣺
������������m��ֵΪ ��
�ڸ��ݱ������ݼ���ϡ������H2SO4������������