��Ŀ����
��2006?�ɶ���ij���������᳧���豸��ª�������¾ɣ��ó�ÿ���ŷŴ�����SO2�ķ����ͺ�H2SO4�����Է�ˮ�����ص����������;������ú̿��ȼ�ϣ�ֻҪ����������꣬�Ը�����ɼ����ƻ���
��1�����������������ԭ��
��2����һ��˵������Ի�����ɵ�Σ����
��3������ij��ѧ����С���������������Ĵ�ʩ������Ϊ������
A�������᳧������� B�����黷��������������
C�������᳧�ų��ķ����е�SO2�������ŷ� D�������;�����ý�����ȼ��
��4��������ʯ�����������᳧�ų������Է�ˮ������ԭ���Ļ�ѧ����ʽ��
��1�����������������ԭ��
�ŷŶ����������
�ŷŶ����������
����2����һ��˵������Ի�����ɵ�Σ����
Ӱ��ũ��������
Ӱ��ũ��������
����3������ij��ѧ����С���������������Ĵ�ʩ������Ϊ������
A
A
��ѡ����ĸ����A�������᳧������� B�����黷��������������
C�������᳧�ų��ķ����е�SO2�������ŷ� D�������;�����ý�����ȼ��
��4��������ʯ�����������᳧�ų������Է�ˮ������ԭ���Ļ�ѧ����ʽ��
Ca��OH��2+H2SO4=CaSO4+2H2O
Ca��OH��2+H2SO4=CaSO4+2H2O
����������1�����������γɵ�ԭ���ǣ���2�����������Σ�����ǣ���3��ֻҪ������������ʹ�����4�����ݷ���ʽ��д�������ǣ�
����⣺��1���������������������γ����ꣻ
��2�������Σ����Ӱ��ũ������������ʴ����ȣ�
��3�������᳧���������Ⱦ��������û�м��٣���A����
��4����Ӧ�����������ƺ����ᣬ������������ƺ�ˮ���ù۲취��ƽ���ɣ�
�ʴ�Ϊ��
��1���ŷŶ������������
��2��Ӱ��ũ����������
��3��A��
��4��Ca��OH��2+H2SO4=CaSO4+2H2O
��2�������Σ����Ӱ��ũ������������ʴ����ȣ�
��3�������᳧���������Ⱦ��������û�м��٣���A����
��4����Ӧ�����������ƺ����ᣬ������������ƺ�ˮ���ù۲취��ƽ���ɣ�
�ʴ�Ϊ��
��1���ŷŶ������������
��2��Ӱ��ũ����������
��3��A��
��4��Ca��OH��2+H2SO4=CaSO4+2H2O
������ͨ���ش���֪���������Ӱ�������ǵ����������������������£�Σ���Ǹ�ʴũ�������Ϥ�˷���ʽ����д������

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ�������������ϵ�д�
�Ƹ�С״Ԫ�������������ϵ�д� ����һ������ܼƻ�ϵ�д�
����һ������ܼƻ�ϵ�д�
�����Ŀ

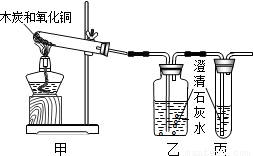 ��2006?�ɶ���ijʵ��������������ͼ����װ����ľ̿��ԭ����ͭ��ʵ���У����ִ���װ�õĵ����ܿڲ����������ݳ�����Ϊ�Ƿ�Ӧ������CO2δ��ʯ��ˮ��ȫ���գ���������װ�ú��������ϱ�װ�ã�����ʵ�飬���ֱ�װ�����Բ����������ݳ�����ʯ��ˮδ�����ǣ���ô�ӱ�װ�����ݳ���������ʲô�أ����������̽���е�������⣮
��2006?�ɶ���ijʵ��������������ͼ����װ����ľ̿��ԭ����ͭ��ʵ���У����ִ���װ�õĵ����ܿڲ����������ݳ�����Ϊ�Ƿ�Ӧ������CO2δ��ʯ��ˮ��ȫ���գ���������װ�ú��������ϱ�װ�ã�����ʵ�飬���ֱ�װ�����Բ����������ݳ�����ʯ��ˮδ�����ǣ���ô�ӱ�װ�����ݳ���������ʲô�أ����������̽���е�������⣮