��Ŀ����
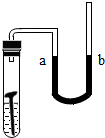
��2011?���죩ʵ��������ƿ��ǩģ������Һ���ֱ�����������������Ϊ8%��NaOH��KOH��Һ��Ϊ�˼�������ƿ��Һ���������������Լ�ƿ�����ϱ�ǩA��B��ʾ����Ȼ��ȡA��B��Һ��20�ˣ��ֱ�����ձ��У���������������Ϊ7.3%��ϡ�����кͣ�����������������ձ�����Һ����������ͼ��ʾ��
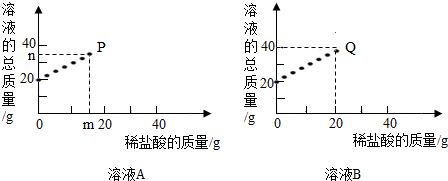
��֪��P��Q������Һ�����ԣ���
��1����ҺB��������
��2�����m��n��ֵ��д��������̣�������һλС������

��֪��P��Q������Һ�����ԣ���
��1����ҺB��������
NaOH
NaOH
��д��ѧʽ������2�����m��n��ֵ��д��������̣�������һλС������
�����������������֪��������������������ƺͺ��������ؽ���ֱ�����ᷴӦ���ʿ��Խ�����Ƿ�Ӧ�Ļ�ѧ����ʽ���㣬���ж���ҺA��B�е����ʼ�m��n��ֵ��
����⣺����P��Q������Һ�����ԣ�����֪����������������������ƺ��������ض���ȫ��Ӧ�������������Ƶ�����Ϊ��20g��8%=1.6g���������ص�����Ϊ��20g��8%=1.6g���ʿ��Ը������ǵ�������ϻ�ѧ����ʽ���м��㣻
������������ȫ��Ӧ�������������Ϊx��
NaOH+HCl�TNaCl+H2O
40 36.5
1.6g x?7.3%
=
��ã�x=20g
������������ȫ��Ӧ�������������Ϊy��
KOH+HCl�TKCl+H2O
56 36.5
1.6g y?7.3%
=
��ã�y=14.3g
��1�����ݼ������֪������������Һ�������������Ϊ20g���ʷ�Ӧ����Һ������Ϊ20g+20g=40g��������ҺB��ͼ���Ǻϣ�������ҺB�е�����Ϊ�������ƣ�
��2�����ݼ������֪����ҺAΪ����������Һ���������������Ϊ14.3g��m=14.3g��n=20g+14.3g=34.3g��
�𣺣�1����ҺB�е�����ΪNaOH��
��2��m��ֵΪ14.3g��n��ֵΪ34.3g��
�ʴ�Ϊ����1��NaOH��
��2��m=14.3g��n=34.3g��
������������ȫ��Ӧ�������������Ϊx��
NaOH+HCl�TNaCl+H2O
40 36.5
1.6g x?7.3%
| 40 |
| 36.5 |
| 1.6g |
| x?7.3% |
��ã�x=20g
������������ȫ��Ӧ�������������Ϊy��
KOH+HCl�TKCl+H2O
56 36.5
1.6g y?7.3%
| 56 |
| 36.5 |
| 1.6g |
| y?7.3% |
��ã�y=14.3g
��1�����ݼ������֪������������Һ�������������Ϊ20g���ʷ�Ӧ����Һ������Ϊ20g+20g=40g��������ҺB��ͼ���Ǻϣ�������ҺB�е�����Ϊ�������ƣ�
��2�����ݼ������֪����ҺAΪ����������Һ���������������Ϊ14.3g��m=14.3g��n=20g+14.3g=34.3g��
�𣺣�1����ҺB�е�����ΪNaOH��
��2��m��ֵΪ14.3g��n��ֵΪ34.3g��
�ʴ�Ϊ����1��NaOH��
��2��m=14.3g��n=34.3g��
������������������ͳ����ļ�ķ�Ӧ�����ܹ���������֮�䷴Ӧ�Ļ�ѧ����ʽ���м��㣬��ס��ѧ����ʽ��NaOH+HCl�TNaCl+H2O��KOH+HCl�TKCl+H2O��

��ϰ��ϵ�д�
�����Ŀ
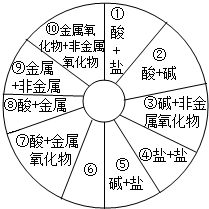 ��2011?���죩���ʡ�������ᡢ��Σ���Щ��ͬ��������֮�䷴Ӧʱ�����������Σ�����ͬѧ��������ͼ��ʾ��֪ʶ����ͼ��
��2011?���죩���ʡ�������ᡢ��Σ���Щ��ͬ��������֮�䷴Ӧʱ�����������Σ�����ͬѧ��������ͼ��ʾ��֪ʶ����ͼ��