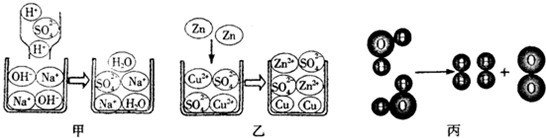
��Ŀ����
��������a��b��С�⣬������ѡһ���������ⶼ�⣬����aС��Ʒ֣�
a����ҵ�ϳ�������ʯ��ʯ�ķ�����ȡ��ʯ�ң����õ���ʯ��һ��Ϊ��ɫ��״���Ӧ�÷dz��㷺����д����
��1����ҵ������ʯ��ʯ��ȡ��ʯ�ҵĻ�ѧ����ʽ��______��
��2����ʯ�ҳ�����ʳƷ������������ʱ�����Ļ�ѧ��Ӧ����ʽ��______��
��3������ʯ����ˮ����20%��ʯ���飬Ϳˢǽ��͵����ܶ���������������������������ĸý�״��Ҫ�������ã����ù��ã���ҪʧЧ����ԭ���ǣ��û�ѧ����ʽ��ʾ��______��
b�����������ճ�������Ӧ����Ϊ�㷺�Ľ����������������գ�
��1�������е�������Ʒ���������⣬���˽�����ϡ�����ȥ����Ʒ��������⣬������������ԣ�������ϡ�����ȥ����Ʒ���������������______ ���û�ѧ����ʽ���ã���
��2�����������е�һЩ�����ܷ�ֹ������ľٴ���______
A�����й��˵ĵ�����ˮϴ������� B�����������õIJ˵����ںڰ���
C�������Ƶ��ſ�����Ϳˢ���� D�������г����������Է���ȡ�ã�
a����ҵ�ϳ�������ʯ��ʯ�ķ�����ȡ��ʯ�ң����õ���ʯ��һ��Ϊ��ɫ��״���Ӧ�÷dz��㷺����д����
��1����ҵ������ʯ��ʯ��ȡ��ʯ�ҵĻ�ѧ����ʽ��______��
��2����ʯ�ҳ�����ʳƷ������������ʱ�����Ļ�ѧ��Ӧ����ʽ��______��
��3������ʯ����ˮ����20%��ʯ���飬Ϳˢǽ��͵����ܶ���������������������������ĸý�״��Ҫ�������ã����ù��ã���ҪʧЧ����ԭ���ǣ��û�ѧ����ʽ��ʾ��______��
b�����������ճ�������Ӧ����Ϊ�㷺�Ľ����������������գ�
��1�������е�������Ʒ���������⣬���˽�����ϡ�����ȥ����Ʒ��������⣬������������ԣ�������ϡ�����ȥ����Ʒ���������������______ ���û�ѧ����ʽ���ã���
��2�����������е�һЩ�����ܷ�ֹ������ľٴ���______
A�����й��˵ĵ�����ˮϴ������� B�����������õIJ˵����ںڰ���
C�������Ƶ��ſ�����Ϳˢ���� D�������г����������Է���ȡ�ã�
a����1������ʯ��ʯ�ᷢ���ֽⷴӦ�������������������̼�����ǹ�ҵ��ȡ������̼��һ�ַ�ʽ����ѧ����ʽΪCaCO3
CaO+CO2����
��2��������������ˮ����ˮ��Ӧ�����������ƣ�������ʳƷ���������ѧ����ʽΪCaO+H2O=Ca��OH��2��
��3����ʯ�ҿ���ˮ��Ӧ�����������ƣ��������ƿ���ɱ�������������������Ϳ����еĶ�����̼��Ӧ����̼��ƣ����ɵ�̼��Ʋ����������ԣ���ѧ����ʽΪCa��OH��2+CO2=CaCO3��+H2O��
b����1�������е�������Ʒ���������⣬���˽�����ϡ�����ȥ����Ʒ��������⣬������������ԣ���Ϊ������ϡ���ᷴӦ����������������������ѧ����ʽΪFe+H2SO4=FeSO4+H2����
��2��A���ú�ʱ��ϴ�����dz��õ�һ�ַ�ֹ�˵�����ķ�������A��ȷ��
B���Ѳ˵�����ںڰ��ĵط���Ϊ�˵������ṩ������������ٲ˵����⣬��B����ȷ��
C��Ϳ����ʹ����ˮ��������������ã���C��ȷ��
D�����г����ڷ������⣬��ӿ����г������⣬��D����
�ʴ�Ϊ��
a����1��CaCO
CaO+CO2������2��CaO+H2O=Ca��OH��2����3��Ca��OH��2+CO2=CaCO3��+H2O��
b����1��Fe+H2SO4=FeSO4+H2������2��AC��
| ||
��2��������������ˮ����ˮ��Ӧ�����������ƣ�������ʳƷ���������ѧ����ʽΪCaO+H2O=Ca��OH��2��
��3����ʯ�ҿ���ˮ��Ӧ�����������ƣ��������ƿ���ɱ�������������������Ϳ����еĶ�����̼��Ӧ����̼��ƣ����ɵ�̼��Ʋ����������ԣ���ѧ����ʽΪCa��OH��2+CO2=CaCO3��+H2O��
b����1�������е�������Ʒ���������⣬���˽�����ϡ�����ȥ����Ʒ��������⣬������������ԣ���Ϊ������ϡ���ᷴӦ����������������������ѧ����ʽΪFe+H2SO4=FeSO4+H2����
��2��A���ú�ʱ��ϴ�����dz��õ�һ�ַ�ֹ�˵�����ķ�������A��ȷ��
B���Ѳ˵�����ںڰ��ĵط���Ϊ�˵������ṩ������������ٲ˵����⣬��B����ȷ��
C��Ϳ����ʹ����ˮ��������������ã���C��ȷ��
D�����г����ڷ������⣬��ӿ����г������⣬��D����
�ʴ�Ϊ��
a����1��CaCO
| ||
b����1��Fe+H2SO4=FeSO4+H2������2��AC��

��ϰ��ϵ�д�
 ����Ӣ��ϵ�д�
����Ӣ��ϵ�д�
�����Ŀ
 33����������a��b��С�⣬��ѡһ���𣬲������ⶼ�⣮�����ⶼ�⣬����aС��Ƿ֣�
33����������a��b��С�⣬��ѡһ���𣬲������ⶼ�⣮�����ⶼ�⣬����aС��Ƿ֣�
 ��������a��b��С�⣬������ѡһ���𣬲������ⶼ�⣮�����ⶼ�⣬����aС��Ʒ֣�
��������a��b��С�⣬������ѡһ���𣬲������ⶼ�⣮�����ⶼ�⣬����aС��Ʒ֣�