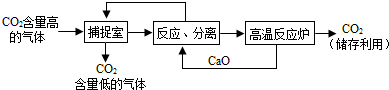
��Ŀ����
��2012?������ģ�⣩��Լ��Դ�ͱ��������Ѿ���Ϊ���ǵĻ������ߣ����ܼ��š�������������̬�н�������������Щ��Ĺ����ص㣮���᳧��������������ǣ��Ѻ������ȼ�գ����ɶ�������������������ڸ��ºʹ���������������������������������ˮ�����������ᣮд�����������ˮ������������Ļ�ѧ����ʽ
ij������Ʒ�к������������ƣ������ⶨ��̼���Ƶ��������������ú�����������ij����ˮ��������ʵ�飺
[ʵ��ԭ��]Na2CO3+H2SO4=Na2SO4+H2O+CO2��
ͨ��ʵ��ⶨ��Ӧ�����Ķ�����̼���������������ԭ��Ʒ��̼���Ƶ��������������̼��������Ʒ�е�����������
[ʵ��װ��]
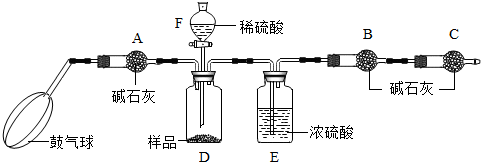
[ʵ�鲽��]
����ͼ����װ�ã���B��C�⣩����������ҩƷ��
�ڳ�������¼B��������m1����������ʱע����B�����ˣ���
�۰������������������Լ1���ӣ�
��������B��C��
�ݴ�Һ©��F�Ļ�������ϡ������ټ���D�кرջ�����
�ް�����������Լ1���ӣ�
�߳�������¼B��������m2����������ʱע����B�����˼�E�Ҷ˵ij��ڣ���
����㣮
��1����֪��ʯ�ҵ���Ҫ�ɷ����������ƺ��������ƣ�������A�������ǣ�
��2��
��3��Eװ�õ�������
��4����ʵ���ܷ�ʡ�Ԣۡ����������裿
��5������ȡ��Ʒ������Ϊ6g����Һ©��F��ʢ��5%ֻ������һ�����ʵij����ˮ���Ƶ�m1Ϊ51.20g��m2Ϊ53.40g����������������λС����
��
��1����Ʒ��̼���Ƶ���������Ϊ���٣�
��2��Dװ�������÷�Ӧ��������Һ���������������Ƕ��٣�
SO3+H2O=H2SO4
SO3+H2O=H2SO4
ij������Ʒ�к������������ƣ������ⶨ��̼���Ƶ��������������ú�����������ij����ˮ��������ʵ�飺
[ʵ��ԭ��]Na2CO3+H2SO4=Na2SO4+H2O+CO2��
ͨ��ʵ��ⶨ��Ӧ�����Ķ�����̼���������������ԭ��Ʒ��̼���Ƶ��������������̼��������Ʒ�е�����������
[ʵ��װ��]

[ʵ�鲽��]
����ͼ����װ�ã���B��C�⣩����������ҩƷ��
�ڳ�������¼B��������m1����������ʱע����B�����ˣ���
�۰������������������Լ1���ӣ�
��������B��C��
�ݴ�Һ©��F�Ļ�������ϡ������ټ���D�кرջ�����
�ް�����������Լ1���ӣ�
�߳�������¼B��������m2����������ʱע����B�����˼�E�Ҷ˵ij��ڣ���
����㣮
��1����֪��ʯ�ҵ���Ҫ�ɷ����������ƺ��������ƣ�������A�������ǣ�
��ֹ�����ж�����̼��ˮ�����ĸ���
��ֹ�����ж�����̼��ˮ�����ĸ���
������ʹ�ⶨ���ƫ����2��
����
����
���ܻ��ܣ���ϡ�������ϡ���ᣬ��Ϊ��������ӷ���
�ӷ���
�ԣ���ʹ���̼���Ƶ���������ƫ��
ƫ��
����ƫ��ƫС�䣬��ͬ������ȥ�������C������̼���Ƶ�������������ƫ��
ƫ��
��3��Eװ�õ�������
��ȥ���ɵĶ�����̼�е�ˮ����
��ȥ���ɵĶ�����̼�е�ˮ����
����4����ʵ���ܷ�ʡ�Ԣۡ����������裿
����
����
�����ܻ��ܣ���ԭ��ֱ���������Ϊ���ų�D�еĿ���
������Ϊ���ų�D�еĿ���
��������Ϊ�˽�D�����ɵĶ�����̼ȫ���ų�
������Ϊ�˽�D�����ɵĶ�����̼ȫ���ų�
����5������ȡ��Ʒ������Ϊ6g����Һ©��F��ʢ��5%ֻ������һ�����ʵij����ˮ���Ƶ�m1Ϊ51.20g��m2Ϊ53.40g����������������λС����
��
��1����Ʒ��̼���Ƶ���������Ϊ���٣�
��2��Dװ�������÷�Ӧ��������Һ���������������Ƕ��٣�
��������1�����ø����Aͨ�������ų�װ��D�еĿ������ܷ�ֹ������ˮ�����Ͷ�����̼�ĸ��Ž��
��2�����������ܻ�ӷ����Ȼ������壬�Ȼ����ܺͼ�ʯ�ҷ�Ӧ�������C�ܷ�ֹ�����еĶ�����̼��ˮ��������B���
��3������Ũ���������ˮ��������ý��
��4������ǰһ�ι�����Ϊ���ų�D�еĿ�����ֹ�����ж�����̼����ĸ��ţ���һ�ι�����Ϊ�˽����ɵĶ�����̼ȫ���ų����
��5���ٸ��ݷ�Ӧǰ��Bװ�����ص�������ϡ�����̼���Ʒ�Ӧ���ɵĶ�����̼�����������̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ���6g��Ʒ��̼���Ƶ����������ɽ�𣮢ڸ��ݷ�Ӧǰ��Bװ�����ص�������ϡ�����̼���Ʒ�Ӧ���ɵĶ�����̼�����������̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ�����Ӧ���ɵ������ƺ����������ʵ�������Ȼ�����ϡ�����������������Ĺ�ʽ���ϡ������Һ�����������������غ㶨�����������������Һ�����������������Ƶ�����������������Һ����������100%���ɽ��
��2�����������ܻ�ӷ����Ȼ������壬�Ȼ����ܺͼ�ʯ�ҷ�Ӧ�������C�ܷ�ֹ�����еĶ�����̼��ˮ��������B���
��3������Ũ���������ˮ��������ý��
��4������ǰһ�ι�����Ϊ���ų�D�еĿ�����ֹ�����ж�����̼����ĸ��ţ���һ�ι�����Ϊ�˽����ɵĶ�����̼ȫ���ų����
��5���ٸ��ݷ�Ӧǰ��Bװ�����ص�������ϡ�����̼���Ʒ�Ӧ���ɵĶ�����̼�����������̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ���6g��Ʒ��̼���Ƶ����������ɽ�𣮢ڸ��ݷ�Ӧǰ��Bװ�����ص�������ϡ�����̼���Ʒ�Ӧ���ɵĶ�����̼�����������̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ�����Ӧ���ɵ������ƺ����������ʵ�������Ȼ�����ϡ�����������������Ĺ�ʽ���ϡ������Һ�����������������غ㶨�����������������Һ�����������������Ƶ�����������������Һ����������100%���ɽ��
����⣺��1�������Aͨ�������ų�װ��D�еĿ������ܷ�ֹ������ˮ�����Ͷ�����̼�ĸ��ţ��ʴ𰸣���ֹ������ˮ�����Ͷ�����̼�ĸ��ţ�
��2�������ܻ�ӷ����Ȼ������壬�Ȼ����ܺͼ�ʯ�ҷ�Ӧ�������C�ܷ�ֹ�����еĶ�����̼��ˮ��������B���ʴ𰸣����ܣ��ӷ��ԣ�ƫ��ƫ��
��3��Ũ���������ˮ��������ã��ʴ𰸣���ȥ���ɵĶ�����̼�е�ˮ������
��4��ǰһ�ι�����Ϊ���ų�D�еĿ�����ֹ�����ж�����̼����ĸ��ţ���һ�ι�����Ϊ�˽����ɵĶ�����̼ȫ���ų����ʴ𰸣����ܣ�������Ϊ���ų�D�еĿ�����������Ϊ�˽�D�����ɵĶ�����̼ȫ���ų���
��5����ϡ���������������Ϊx�����ɵ������Ƶ�����Ϊy����Ʒ��̼���Ƶ�����Ϊz��Bװ�����ص���������̼���ƺ�ϡ���ᷴӦ���ɵĶ�����̼������Ϊ��53.40g-51.20g=2.2g
Na2CO3+H2SO4=Na2SO4+H2O+CO2��
106 98 142 44
z x y 2.2g
=
x=4.90g
=
y=7.10g
=
z=5.3g
��1����Ʒ��̼���Ƶ���������Ϊ��
��100%=88.3%
��2������ϡ���������Ϊ
=98g
������������Һ������Ϊ98g+5.3g-2.2g=101.1g
������������Һ��������������Ϊ��
��100%=7.02%
����Ʒ��̼���Ƶ���������Ϊ88.3%��������Һ�������Ƶ���������Ϊ7.02%
��2�������ܻ�ӷ����Ȼ������壬�Ȼ����ܺͼ�ʯ�ҷ�Ӧ�������C�ܷ�ֹ�����еĶ�����̼��ˮ��������B���ʴ𰸣����ܣ��ӷ��ԣ�ƫ��ƫ��
��3��Ũ���������ˮ��������ã��ʴ𰸣���ȥ���ɵĶ�����̼�е�ˮ������
��4��ǰһ�ι�����Ϊ���ų�D�еĿ�����ֹ�����ж�����̼����ĸ��ţ���һ�ι�����Ϊ�˽����ɵĶ�����̼ȫ���ų����ʴ𰸣����ܣ�������Ϊ���ų�D�еĿ�����������Ϊ�˽�D�����ɵĶ�����̼ȫ���ų���
��5����ϡ���������������Ϊx�����ɵ������Ƶ�����Ϊy����Ʒ��̼���Ƶ�����Ϊz��Bװ�����ص���������̼���ƺ�ϡ���ᷴӦ���ɵĶ�����̼������Ϊ��53.40g-51.20g=2.2g
Na2CO3+H2SO4=Na2SO4+H2O+CO2��
106 98 142 44
z x y 2.2g
| 98 |
| x |
| 44 |
| 2.2g |
x=4.90g
| 142 |
| y |
| 44 |
| 2.2g |
y=7.10g
| 106 |
| z |
| 44 |
| 2.2g |
z=5.3g
��1����Ʒ��̼���Ƶ���������Ϊ��
| 5.3g |
| 6g |
��2������ϡ���������Ϊ
| 4.90g |
| 0.05 |
������������Һ������Ϊ98g+5.3g-2.2g=101.1g
������������Һ��������������Ϊ��
| 7.1g |
| 101.1g |
����Ʒ��̼���Ƶ���������Ϊ88.3%��������Һ�������Ƶ���������Ϊ7.02%
����������ͨ��ʵ��̽�����̿����˼�ʯ�Ҽ�����ˮ���ܺͶ�����̼��Ӧ��Ũ���������ˮ�ԡ�̼���ƺ�ϡ���ᷴӦ���������ơ�ˮ�Ͷ�����̼�����ҿ����˸���̼���ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽ�Ľ����Һ�������������ļ��㣬�ѶȽϴ�ֻҪ�������ÿ���IJ���Ŀ�IJ��ܽ����⣮

��ϰ��ϵ�д�
�����Ŀ
 ��2012?�����ж�ģ����ͼ��̽���кͷ�Ӧ�е������仯��װ�ã�����U�ι��������˵�Һ��߶�һ�£��������ᣬ�۲����������й�˵������ȷ���ǣ�������
��2012?�����ж�ģ����ͼ��̽���кͷ�Ӧ�е������仯��װ�ã�����U�ι��������˵�Һ��߶�һ�£��������ᣬ�۲����������й�˵������ȷ���ǣ�������