��Ŀ����
ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ���1���ס�����ͬѧ������������װ�ü�������ԣ���ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ���
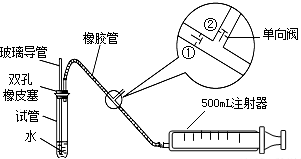
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ�
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�������������
��2�����Թ��м���0��00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
��4��ͨ���ⶨ�����㣬���жϳ�����ص�Ŀ�����S02�ĺ�������
| ���� | ��һС�� | �ڶ�С�� |
| ����ʱ�� | 20���� | 21���� |
| �������� | 100 | 130 |
| SO2������mg/m3 |
����ʵ������У��軺���鶯������Ŀ����
���жϸõص�Ŀ�����SO2��������
������Ū��װ�������Եļ������Ҫ��֤װ�����γ�һ���ܱյ���ϵ�������ܱ���ϵ�ڵ�ѹǿ�ı仯�����ڵ�ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ���˼���װ�õ�������ʱ��Ϊʹ�γ�һ���ܱյĿռ䣬Ӧ��ע�����Ļ�����ʹװ���ڵ�ѹǿ��С���Ӷ��ڽ�û��ˮ�еIJ������ܿڻῴ��������ð��������SO2+I2+2H2O=H2SO4+2HI�������ʵ�ǡ����ȫ��Ӧʱ�����������ɵ���ɫ�ͻ���ʧ���Ӷ����ݲμӷ�Ӧ�ĵ�����������������������������Ӷ�ȷ���˵ؿ����ж�������ĺ�����
����⣺��1�����ڲ��������������ͬ��û�γ��ܱյ���ϵ������ͬѧ��Ȼ��������עˮ��û��ʹ��ѹǿ�����仯���������ͬѧ��û�������װ�õ��������Ƿ���ã��ʴ�Ϊ�����ܣ�
��Ҫ�����װ�õ������ԣ��������ⵥ�����ã�����ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ����Ӧ������ע�����Ļ����������Թ���ѹǿ��С����������û��ˮ�еIJ����ܿڴ�������ð����˵�����������ã��ʴ�Ϊ��������������ע�����Ļ�������û��ˮ�еIJ������ܿ�������ð��
��2�����ṩ�Ļ�ѧ����ʽ���ѿ���������Ӧ��ǡ����ȫ��Ӧʱ����Һ�в����е��ʵ⣬��Һ����ɫ�ͻ���ʧ���ʴ�Ϊ��������ɫ
��3��������ȡ�õ�ˮ��һ���ģ��������ʵ���в��뷴Ӧ�Ķ������������һ���࣬Ϊ��ʹ��������������ȫ�����գ���˳�������ʱҪ�����ؽ��У����ݻ�ѧ����ʽ���㣬����뷴Ӧ�Ķ������������ΪX��
SO2 +I2 +2H2O=H2SO4 +2HI
64 254
X 10g��0.00127%
=
X=
=0.000032g=0.032mg
��һ�飺
=0.64mg/m3
�ڶ��飺
=0.49mg/m3
�ʴ�Ϊ����0.64��0.49��Ŀ�����ö�����������ַ�Ӧ����ʹ�����ж�����������գ���ƫС
����������һ���������ʹ��죬��ɿ�����SO2���ˮδ��ַ�Ӧ�������ϴ����
��Ҫ�����װ�õ������ԣ��������ⵥ�����ã�����ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ����Ӧ������ע�����Ļ����������Թ���ѹǿ��С����������û��ˮ�еIJ����ܿڴ�������ð����˵�����������ã��ʴ�Ϊ��������������ע�����Ļ�������û��ˮ�еIJ������ܿ�������ð��
��2�����ṩ�Ļ�ѧ����ʽ���ѿ���������Ӧ��ǡ����ȫ��Ӧʱ����Һ�в����е��ʵ⣬��Һ����ɫ�ͻ���ʧ���ʴ�Ϊ��������ɫ
��3��������ȡ�õ�ˮ��һ���ģ��������ʵ���в��뷴Ӧ�Ķ������������һ���࣬Ϊ��ʹ��������������ȫ�����գ���˳�������ʱҪ�����ؽ��У����ݻ�ѧ����ʽ���㣬����뷴Ӧ�Ķ������������ΪX��
SO2 +I2 +2H2O=H2SO4 +2HI
64 254
X 10g��0.00127%
| 64 |
| 254 |
| X |
| 10g��0.00127% |
X=
| 64��10g��0.00127% |
| 254 |
��һ�飺
| 0.032mg |
| 500��100��10-6m3 |
�ڶ��飺
| 0.032mg |
| 500��130��10-6m3 |
�ʴ�Ϊ����0.64��0.49��Ŀ�����ö�����������ַ�Ӧ����ʹ�����ж�����������գ���ƫС
����������һ���������ʹ��죬��ɿ�����SO2���ˮδ��ַ�Ӧ�������ϴ����
��������Ҫ�Կ����ж�������ĺ���������̽����������һ������ȣ��ѶȽϴ���Ҫ����ѧ���������⡢��������������

��ϰ��ϵ�д�
�����Ŀ
SO2�Ǵ�����Ⱦ��֮һ��Ϊ���Բⶨ��Χ������SO2�ĺ�����ij����С�����������ʵ��װ�ã�

ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ�
��ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ� �����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ� �����ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ������������� ������ ����ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0.00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ�� ɫ��Ϊ ʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
����ʵ������У��軺���鶯������Ŀ���� �������ٳ�������ⶨ�Ľ���� ����ƫ��ƫС����Ӱ�죩��
���жϸõص�Ŀ�����SO2�������� �������֣������� �����һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩ ��

ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ�
��ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ�
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ�
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�������������
��2�����Թ��м���0.00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
| ���� | ��һС�� | �ڶ�С�� |
| ����ʱ�� | 20���� | 21���� |
| �������� | 100 | 130 |
| SO2������mg/m3 |
���жϸõص�Ŀ�����SO2��������
SO2�Ǵ�����Ⱦ��֮һ��Ϊ���Բⶨ��Χ������SO2�ĺ�����ij����С�����������ʵ��װ�ã�
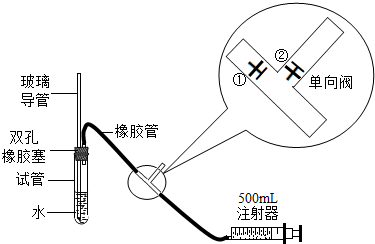
ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ�
��ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ�______�����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ�______�����ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�������������______������______����ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0.00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��______ɫ��Ϊ______ʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
| ���� | ��һС�� | �ڶ�С�� |
| ����ʱ�� | 20���� | 21���� |
| �������� | 100 | 130 |
| SO2������mg/m3 |
���жϸõص�Ŀ�����SO2��������______�������֣�������______�����һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩______��
SO2�Ǵ�����Ⱦ��֮һ��Ϊ���Բⶨ��Χ������SO2�ĺ�����ij����С�����������ʵ��װ�ã�

ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ�
��ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ�______�����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ�______�����ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�������������______������______����ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0.00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��______ɫ��Ϊ______ʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
����ʵ������У��軺���鶯������Ŀ����______�������ٳ�������ⶨ�Ľ����______����ƫ��ƫС����Ӱ�죩��
���жϸõص�Ŀ�����SO2��������______�������֣�������______�����һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩______��

ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ�
��ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ�______�����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ�______�����ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�������������______������______����ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0.00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��______ɫ��Ϊ______ʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
| ���� | ��һС�� | �ڶ�С�� |
| ����ʱ�� | 20���� | 21���� |
| �������� | 100 | 130 |
| SO2������mg/m3 |
���жϸõص�Ŀ�����SO2��������______�������֣�������______�����һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩______��
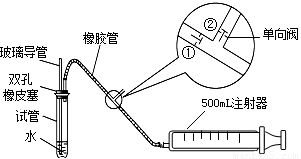
ע������ԭ��˵������ע�����ƻ���ʱ���ٹرգ��ڴ���ע����������ʱ���ٴ��ڹرգ�
��1���ס�����ͬѧ������������װ�ü�������ԣ���ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ��𣺣����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ������ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�����������������������ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0��00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��ɫ��Ϊʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
��4��ͨ���ⶨ�����㣬���жϳ�����ص�Ŀ�����S02�ĺ������ڱ������3�����о�
��һС�飺���ڶ�С�飺��
����ʵ������У��軺���鶯������Ŀ���ǣ������ٳ�������ⶨ�Ľ��������ƫ��ƫС����Ӱ�죩��
���жϸõص�Ŀ�����SO2�������ڣ������֣������������һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩��
��1���ס�����ͬѧ������������װ�ü�������ԣ���ͬѧ��������ס�Թܣ������Թܣ����۲쵼�����Ƿ���Һ�������������Ƿ��ܣ��𣺣����ܻ��ܣ���
��ͬѧ���ȴӲ����������Թ���ע��ˮ���۲쵼����Һ�����Թ���Һ���Ƿ��γɸ߶Ȳ����һ��ʱ��۲�Һ����Ƿ�仯�ķ������Ƿ��ܣ������ܻ��ܣ���
���������ַ��������ܣ���˼���·������������пհף����������У��������пհף��������Թ���װ������ˮ����֤�������ܵ��¶˽�û��ˮ�У���Ȼ�����������������������ʵ������֤����װ�õ����������ã�
��2�����Թ��м���0��00127%�ĵ�ˮ10g��������������ˮϡ�ͺ��ټ���2��3�ε�����Һ�����ó���ҺA���ⶨָ���ص������SO2�ĺ���ʱ������ע�����Ļ��������������Թ��з����Ļ�ѧ��Ӧ����ʽΪSO2+I2+2H2O=H2SO4+2HI��A��Һ��ɫ��Ϊʱ������Ӧǡ����ȫ���У���ʱӦֹͣ������
��3���ҹ����������������ж�ÿ�ο��������ⶨ��SO2�����Ũ����ֵ��mg/m3����һ������0.15����������0.50����������0.70���ÿ���С��ֳɵ�һ�С��͵ڶ��С�飬ʹ����ͬ��ʵ��װ�ú���ҺA����ͬһ�ص㡢ͬʱ����������SO2�ĺ���������Ӧǡ����ȫ���У���¼����ʱ��ʹ������£��ٶ�ÿ�γ���500mL�����뽫�±���д����������ʱ����2λ��Ч���֣���
��4��ͨ���ⶨ�����㣬���жϳ�����ص�Ŀ�����S02�ĺ������ڱ������3�����о�
| ���� | ��һС�� | �ڶ�С�� |
| ����ʱ�� | 20���� | 21���� |
| �������� | 100 | 130 |
| SO2������mg/m3 |
����ʵ������У��軺���鶯������Ŀ���ǣ������ٳ�������ⶨ�Ľ��������ƫ��ƫС����Ӱ�죩��
���жϸõص�Ŀ�����SO2�������ڣ������֣������������һ���ڶ�����С��IJⶨ���ȷ����һС��ʵ���������ϴ�ƫ���ԭ���ǣ�����С������ҩƷ��װ�þ������⣩��
