��Ŀ����
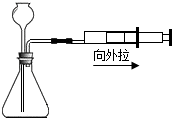
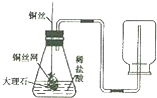
����ͼ��ʾ����ƿ��ʢ������Y����Һ©����ʢ��Һ��X��U����ʢ�к�īˮ�����ڹ۲죩����ʼʱa��b����ͬһˮƽ�ߣ�����X������ƿ��ʱ������ش��������⣺
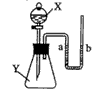
(1)��XΪϡ���ᣬYΪ��ʯ�ң���U����aҺ�� ������ڡ��������ڡ����ڡ�����ͬ)bҺ�棻
(2)��XΪˮ��YΪ����粒��壬��U����aҺ�� bҺ�棻
(3)��XΪ����������Һ��YΪ������̼���壬��U�ι���aҺ�� bҺ�棬��Ӧ�Ļ�ѧ����ʽ�� ��
(2)��XΪˮ��YΪ����粒��壬��U����aҺ�� bҺ�棻
(3)��XΪ����������Һ��YΪ������̼���壬��U�ι���aҺ�� bҺ�棬��Ӧ�Ļ�ѧ����ʽ�� ��
��1������
��2������
��3�����ڣ�2NaOH + CO2===Na2CO3 + H2O
��2������
��3�����ڣ�2NaOH + CO2===Na2CO3 + H2O

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
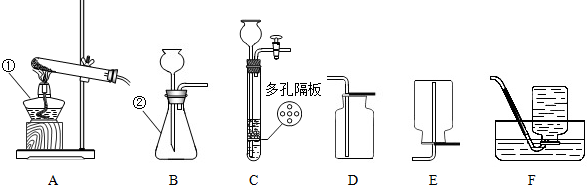
�����Ŀ
 17������ͼ��ʾ��A��B�ֱ�Ϊ���ܱ�װ�ã�װ������ʢ������ͼ�б�ʾ����ʱ��װ�õ�������ȫ��ͬ���Իش��������⣺
17������ͼ��ʾ��A��B�ֱ�Ϊ���ܱ�װ�ã�װ������ʢ������ͼ�б�ʾ����ʱ��װ�õ�������ȫ��ͬ���Իش��������⣺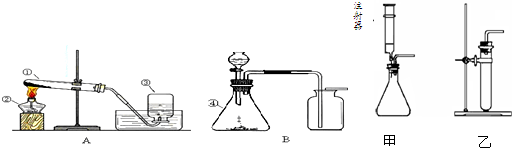
 ����ѧϰ��ʵ������ȡO2��CO2�ķ������������ܹ��ش��������⣺
����ѧϰ��ʵ������ȡO2��CO2�ķ������������ܹ��ش��������⣺