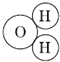
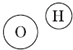��Ŀ����
������з�Ӧ�Ļ�ѧ����ʽ����ע��������Ӧ���ͣ�
��1������������ȼ�� �� ��Ӧ��
��2�����ˮ �� ��Ӧ��
��3��þ���ڿ�����ȼ�� �� ��Ӧ��
��4��һ����̼ȼ�� �� ��Ӧ��
��5����˫��ˮ��ȡ���� �� ��Ӧ��
��6�����ȸ������ �� ��Ӧ��
��7��������̼ͨ�����ʯ��ˮ ��
��8��ʵ�����ƶ�����̼ ��
��9�����飨C21H30O2���ڿ�������ȫȼ�����ɶ�����̼��ˮ ��
��1������������ȼ��
��2�����ˮ
��3��þ���ڿ�����ȼ��
��4��һ����̼ȼ��
��5����˫��ˮ��ȡ����
��6�����ȸ������
��7��������̼ͨ�����ʯ��ˮ
��8��ʵ�����ƶ�����̼
��9�����飨C21H30O2���ڿ�������ȫȼ�����ɶ�����̼��ˮ
���㣺��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,��Ӧ���͵��ж�
ר�⣺��ѧ����������غ㶨��
���������ݷ�Ӧ�������ͷ�Ӧ�����������غ㶨�ɵ�����ԭ��д����ȷ�Ļ�ѧ����ʽ��
����⣺�����ֻ�����������������һ�����ʵķ�Ӧ�л��Ϸ�Ӧ����һ�������������ֻ����������������ʵķ�Ӧ�зֽⷴӦ��
��1������������ȼ�ջ�ѧ����ʽΪ��3Fe+2O2
Fe3O4���ǻ��Ϸ�Ӧ��
��2�����ˮ��ѧ����ʽΪ��2H2O
2H2��+O2�����ǷֽⷴӦ��
��3��þ���ڿ�����ȼ�ջ�ѧ����ʽΪ��2Mg+O2
2MgO���ǻ��Ϸ�Ӧ��
��4��һ����̼ȼ�ջ�ѧ����ʽΪ��2CO+O2
2CO2���ǻ��Ϸ�Ӧ��
��5����˫��ˮ��ȡ������ѧ����ʽΪ��2H2O2
2H2O+O2�����ǷֽⷴӦ��
��6�����ȸ�����ػ�ѧ����ʽΪ��2KMnO4
K2MnO4+MnO2+O2�����ǷֽⷴӦ��
��7��������̼ͨ�����ʯ��ˮ��ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
��8��ʵ�����ƶ�����̼��ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��9�����飨C21H30O2���ڿ�������ȫȼ�����ɶ�����̼��ˮ��ѧ����ʽΪ��2C21H30O2+55O2
42CO2+30H2O��
�ʴ�Ϊ����1��3Fe+2O2
Fe3O4�� ����
��2��2H2O
2H2��+O2���� �ֽ�
��3��2Mg+O2
2MgO�� ����
��4��2CO+O2
2CO2�� ����
��5��2H2O2
2H2O+O2���� �ֽ�
��6��2KMnO4
K2MnO4+MnO2+O2���� �ֽ�
��7��CO2+Ca��OH��2�TCaCO3��+H2O
��8��CaCO3+2HCl�TCaCl2+H2O+CO2��
��9��2C21H30O2+55O2
42CO2+30H2O��
��1������������ȼ�ջ�ѧ����ʽΪ��3Fe+2O2
| ||
��2�����ˮ��ѧ����ʽΪ��2H2O
| ||
��3��þ���ڿ�����ȼ�ջ�ѧ����ʽΪ��2Mg+O2
| ||
��4��һ����̼ȼ�ջ�ѧ����ʽΪ��2CO+O2
| ||
��5����˫��ˮ��ȡ������ѧ����ʽΪ��2H2O2
| ||
��6�����ȸ�����ػ�ѧ����ʽΪ��2KMnO4
| ||
��7��������̼ͨ�����ʯ��ˮ��ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
��8��ʵ�����ƶ�����̼��ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��9�����飨C21H30O2���ڿ�������ȫȼ�����ɶ�����̼��ˮ��ѧ����ʽΪ��2C21H30O2+55O2
| ||
�ʴ�Ϊ����1��3Fe+2O2
| ||
��2��2H2O
| ||
��3��2Mg+O2
| ||
��4��2CO+O2
| ||
��5��2H2O2
| ||
��6��2KMnO4
| ||
��7��CO2+Ca��OH��2�TCaCO3��+H2O
��8��CaCO3+2HCl�TCaCl2+H2O+CO2��
��9��2C21H30O2+55O2
| ||
�����������ѶȲ�����ѧ�����ݷ�Ӧԭ����д��ѧ����ʽ����������ѧ����ʽ��д�������ֵĴ����в����Ͽ���ʵ�������������غ㶨�ɡ���д������������ŵȣ�

��ϰ��ϵ�д�
�����Ŀ
���и������ʣ�ǰ���ǻ���������ǻ������ǣ�������
| A�������������Ͻ� |
| B������������ |
| C������ʯ��ʯ��ʯ |
| D��������̼���ɱ� |

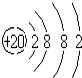 ������ͼ��ʾ��ԭ�ӽṹʾ��ͼ��
������ͼ��ʾ��ԭ�ӽṹʾ��ͼ��