��Ŀ����
��1��������������ѡ����ʵ�����������Ӧ�ĺ����ϣ�
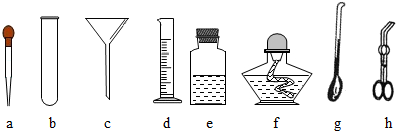
����������
�ڹ�����
����ȡ25.0mLˮ�õ�
�ܿ���ֱ�Ӽ��ȵIJ���������
��2����ͼ�ǻ�ѧʵ���г��õļ���װ�ã�
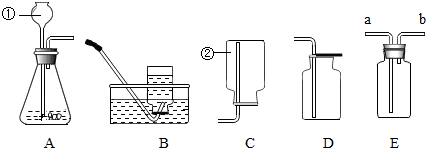
��ش��������⣺
����������A��Dװ�õ���Ͽ�����ȡ��һ�������� ��д��ʵ������ȡ������Ļ�ѧ��Ӧ����ʽ ��
��ijͬѧ���ռ�һ���ܶȱȿ���С�����壬��Ϊ��Eװ�ÿ��ԣ��������ͬѧӦ���ʹ��Eװ�ã� ��

����������
�ڹ�����
����ȡ25.0mLˮ�õ�
�ܿ���ֱ�Ӽ��ȵIJ���������
��2����ͼ�ǻ�ѧʵ���г��õļ���װ�ã�

��ش��������⣺
����������A��Dװ�õ���Ͽ�����ȡ��һ��������
��ijͬѧ���ռ�һ���ܶȱȿ���С�����壬��Ϊ��Eװ�ÿ��ԣ��������ͬѧӦ���ʹ��Eװ�ã�
���㣺�������������ƺ�ѡ��,��������ķ���װ�ú��ռ�װ����ѡȡ����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺������������ѧʵ���������,���������ʵ�����Ʒ������顢�����뾻��
��������1�����ݳ������������ƺ���;����ѡ��
��2��AD��Ͼͷ�Ӧ����˵װ��A���ڹ�Һ�����ȵķ�Ӧ����D�ʺ������ܶȴ��ڿ������ռ���
װ��E���������ռ�װ�ã��ռ�����ʱ����������ܶ�С�ڿ������ܶ�ʱ����b�˽������������ڽ�����ƿ��ԭ�е������ų�ƿ�⣮
��2��AD��Ͼͷ�Ӧ����˵װ��A���ڹ�Һ�����ȵķ�Ӧ����D�ʺ������ܶȴ��ڿ������ռ���
װ��E���������ռ�װ�ã��ռ�����ʱ����������ܶ�С�ڿ������ܶ�ʱ����b�˽������������ڽ�����ƿ��ԭ�е������ų�ƿ�⣮
����⣺��1������������ϸ��ƿ�У�
�ڹ�����©����
����ȡ25.0mLˮ�õ���Ͳ�ͽ�ͷ�ιܣ�
�ܿ���ֱ�Ӽ��ȵIJ����������Թܣ�
��2��AӦ���ڹ�Һ��Ӧ�ķ���װ�ã���װ��B���ڹ�Һ����Ҫ���ȵķ�Ӧ��C�����ܶ�С�ڿ����������ռ�װ�ã�D�����ܶȴ��ڿ����������ռ���ʵ����ͨ����MnO2���������������ù���������ȡ����ʱ�������ʺ�ͬʱ����AD����ȡ�������������������̼��
װ��E���������ռ�װ�ã��ռ�����ʱ����������ܶ�С�ڿ������ܶ�ʱ����b�˽������������ڽ�����ƿ��ԭ�е������ų�ƿ�⣮
�ʴ�Ϊ����1��e��c��ad��b����2����CO2��CaCO3+2HCl=CaCl2+H2O+CO2�����������b�˽��룮
�ڹ�����©����
����ȡ25.0mLˮ�õ���Ͳ�ͽ�ͷ�ιܣ�
�ܿ���ֱ�Ӽ��ȵIJ����������Թܣ�
��2��AӦ���ڹ�Һ��Ӧ�ķ���װ�ã���װ��B���ڹ�Һ����Ҫ���ȵķ�Ӧ��C�����ܶ�С�ڿ����������ռ�װ�ã�D�����ܶȴ��ڿ����������ռ���ʵ����ͨ����MnO2���������������ù���������ȡ����ʱ�������ʺ�ͬʱ����AD����ȡ�������������������̼��
װ��E���������ռ�װ�ã��ռ�����ʱ����������ܶ�С�ڿ������ܶ�ʱ����b�˽������������ڽ�����ƿ��ԭ�е������ų�ƿ�⣮
�ʴ�Ϊ����1��e��c��ad��b����2����CO2��CaCO3+2HCl=CaCl2+H2O+CO2�����������b�˽��룮
������������Ҫ���鳣��ʵ�����������ü�����������������̼��������ʵ��װ���Ʒ���ԭ������ѧ����ʽ����д��ʵ����������ȷʹ�õ�֪ʶ��������֪��

��ϰ��ϵ�д�
�����Ŀ
 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ����ͼ�ش�
��ͼ��a��b��c�������ʵ��ܽ�����ߣ����ͼ�ش�