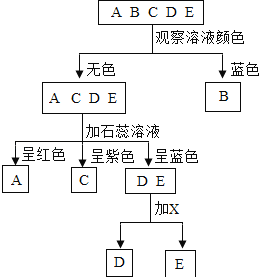
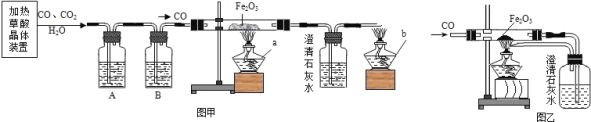
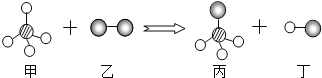
��Ŀ����
����Ŀ��ijѧУ��ѧϰС��Ե��ص�ʯ��ʯ�������е��飬�ⶨʯ��ʯ��̼��Ƶ��������������õķ������£�ȡ��ʯ��ʯ��Ʒ16g����80gϡ������Ĵμ��룬���������������ݼ��±�����֪ʯ��ʯ��Ʒ�к��еĶ�����������ʲ�����ˮ������ϡ���ᷴӦ��������㣺
��� | ����ϡ���������/g | ʣ����������/g |
��1�� | 20 | 11 |
��2�� | 20 | 6 |
��3�� | 20 | 2.8 |
��4�� | 20 | n |
��1���ϱ���n����ֵΪ ��
��2����Ʒ��̼��Ƶ����������� ��
��3�������������ʵ�����������
���𰸡���1��2.8����2��82.5%��
��3�����������ʵ���������Ϊ18.25%��
�����������һ�β��뷴Ӧ��HCl����Ϊx��
CaCO3+2HCl=CaCl2+H2O+CO2��
100 73
5g x
![]()
��֮�ã�x=3.65g��
���������ʵ���������Ϊ��![]() ��100%=18.25%��
��100%=18.25%��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ