��Ŀ����
ij������Ʒ�г�NaCl���������������MgCl2��Ϊ�˲ⶨ�ô�����Ʒ��NaCl��������������������ʵ�飺ȡ��Ʒ60.0g��ȫ����ˮ���200.0g��Һ�����������μ�10%������������Һ��ʵ����������ɳ�����μ�����������Һ��������ϵ��ͼ��ʾ������������Ϣ����������⣺

��1����ȫ��Ӧʱ����������þ������Ϊ______g��
��2���ô�����Ʒ���Ȼ��Ƶ���������______����д��������̣������ȷ��0.1%����ͬ��
 �óɼ�1��1��ĩ���100��ϵ�д�
�óɼ�1��1��ĩ���100��ϵ�д� ��״Ԫ���źþ�ϵ�д�
��״Ԫ���źþ�ϵ�д��ӹ�������������ȡ�赥�ʣ���Ҫ��ѧ��Ӧ���£�
�ֹ����ȡ��SiO2��ʯӢɰ��+2C����̿�� Si���ֹ裩+2CO��
Si���ֹ裩+2CO��
�ɴֹ��ƴ��裨���÷�������Si���֣�+2Cl2 SiCl4 SiCl4+2H2
SiCl4 SiCl4+2H2 Si������+4HCl
Si������+4HCl
��1������������Ϣ���벹ȫ��ȡ����ļ�������ͼ��________ ��

��2����������֪ʶ��������Ϣ�������ܽ���ɣ�
������û���Ӧ��ͨʽ�����ʣ�I��+�����I��=���ʣ���+________ ��
��������
���ʣ�I�� | ���ʣ��� | ��Ӧ���û���Ӧ�Ļ�ѧ����ʽ����һ���� | |
A | ���� | �ǽ��� | Fe+2HCl=FeCl2+H2�� |
B | ���� | ���� | ________��[���ʣ�I��Ϊͭ] |
C | ________ | ________ | ________ |
D | ________ | ���� | H2+CuO |


 2CO2+2H2O��X�Ļ�ѧʽΪ��������
2CO2+2H2O��X�Ļ�ѧʽΪ�������� Cu+H2O
Cu+H2O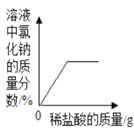 ��һ����NaOH��Һ�еμ�ϡ����
��һ����NaOH��Һ�еμ�ϡ���� ��H2SO4��CuSO4�Ļ����Һ�еμӹ���NaOH��Һ
��H2SO4��CuSO4�Ļ����Һ�еμӹ���NaOH��Һ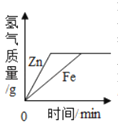 ��������п�ۡ������зֱ�������������Ũ�ȵ�ϡ����
��������п�ۡ������зֱ�������������Ũ�ȵ�ϡ����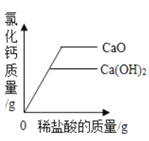 ��������������ơ����������зֱ���������������ϡ����������
��������������ơ����������зֱ���������������ϡ����������