��Ŀ����
�±���ij�����������ɷ֣��ݱ��ش��������⡣

(1)���������������������ͬ������������___________���ж�������_________��
(2)�������������з�������������̵��������������ɷ���ε�����_________��������___________________��
(3)���������������ڹ۲������̬��Ӧ���ӵijɷ���____________��
(4)�ڳ����̼Դ����Դ�����κ�ˮ�������£�ijЩ������Ȼ������������������Ҫ��������������_____________������ѡ�������ڸ������ʵ���________��(������ѡ��)
A��ijЩά���� B��ijЩ������ C��ø D������
(2)�������������з�������������̵��������������ɷ���ε�����_________��������___________________��
(3)���������������ڹ۲������̬��Ӧ���ӵijɷ���____________��
(4)�ڳ����̼Դ����Դ�����κ�ˮ�������£�ijЩ������Ȼ������������������Ҫ��������������_____________������ѡ�������ڸ������ʵ���________��(������ѡ��)
A��ijЩά���� B��ijЩ������ C��ø D������
(1)���������л�̼Դ
(2)ȥ���������Ģڳɷ֣������л�̼Դ�������̵��������ÿ����еĵ�����Ϊ��Դ�������������̵������������������л�������л�̼Դ��
(3)��֬
(4)�������ӣ�AB
(2)ȥ���������Ģڳɷ֣������л�̼Դ�������̵��������ÿ����еĵ�����Ϊ��Դ�������������̵������������������л�������л�̼Դ��
(3)��֬
(4)�������ӣ�AB

��ϰ��ϵ�д�
�����Ŀ
��ÿ��1�֣���14�֣�
����ͻ�������ù�����ڻ����������������ʵ��İ�������������������������ͣ����������������������ͺ�ɫ���ù��ͻ����a��ͻ����b����������ʵ�飺
ʵ��һ������������ͻ����ֱ����������6���������ϣ�����ͻ���겻����l��3��5��
����������������2��4��6�������϶����������������ijɷ����±���ʾ����ͻ����a��
b��Ҫ����A��J�е����������������������
| �������� | �����ɷ� | ���ӵİ��������� |
| �� | | �� |
| �� | | �����¡��á��� |
| �� | ���������� | �¡��š��ơ��� |
| �� | �á��ơ��ȡ��� | |
| �� | �ġ��ǡ��ɡ��� | |
| �� | �����ʶ��� |
��ע��������������Ұ���;��������������������
ʵ������ٶ�ͻ����a��b����İ�����Ϊ�أ���Ұ�������ڣ�����ͼ��ʾ��;���ϳɣأ���Ӧ�١��ڡ��۷ֱ��ɲ�ͬ��ø�����������ӹϰ���ʱ��ͻ����a��������������������������������ӹϰ��ỹ�����ᣬͻ����b������������
����������Ӧ�� ��Ӧ�� ��Ӧ��


 ���� �������� �ϰ��� ����������X
���� �������� �ϰ��� ����������Xͻ����a����������ԭ���������������裬ͻ����b�����������������裬��Ӧ����ĸ���ԭ��������������������������������
ʵ������������Y��������Ҫ���϶�İ����ᣬΪʹ���ù���ڴ�л�����в�������İ�����Y��������ͼ�жϡ�
ø�� ø�� ø��


 ���� ���� �ϰ��� ������X
���� ���� �ϰ��� ������X
ø��
������Y
Ӧ�ö����ù�п��������������ϳɵĻ�������ձ��ƻ����ձ�֮ǰ���Ŵ���Ϣ�Ĵ���;��������������������������������������
���ش������й���������⣺
������ͼһΪijϸ���ϳ�ij�ְ�����Ĵ�л����ʾ��ͼ�����ұ�����ʾ�м�����
|

�������л�ĵ���������ø����________ø����������ϸ���л��۹��࣬������ø��Ļ��ԣ����ֵ��ڷ�ʽ�����������������������������ص㡣
��2���±���ij�����������ɷ֡�����ݴ˱��ش��������⣺
| ��� | �� | �� | �� | �� | �� | �� | �� |
| �ɷ� | S | ��NH4��2SO4 | K2HPO4 | MgSO4 | FeSO4 | CaCl2 | H2O |
| ���� | 10g | 0��4g | 4��0g | 9��25g | 0��5g | 0��5g | 100mL |
��ijͬѧ�ô��������ֱ���������ͽ�ĸ������������װ�÷��ڹ��������������ⶨ����Һ������ͽ�ĸ����������������Ƴ�����ͼ������ͬ���������ͷ�����������������������������ԭ����������������������������

��Ŀǰͨ�������̿����������������ȵ��ء�ͼ����ʾ�������ȵ��ػ���Ĵ˾��������ߣ�����BC������________�ڣ�����AC�δ˾���Ⱥ�����仯��________���������˾���л����ȵ��أ�����ʱ���൱�����ߵ�________�Σ���ϸ�����ֶ�
����̬����������ʱ���˾�Ⱥ���������뵽ͼ�е�________�Ρ�
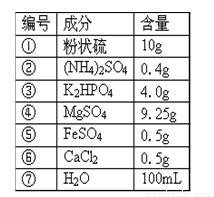
�±���ij�����������ɷ֣���ݴ˻ش�
| ��� | �ɷ� | ���� |
| �� | ��״�� | 10g |
| �� | ��NH4��2SO4 | 0.4g |
| �� | K2HPO4 | 4.0g |
| �� | MgSO4 | 9.25g |
| �� | FeSO4 | 0.5g |
| �� | CaCl2 | 0.5g |
| �� | H2O | 100ml |
��1������������������������������ ������ȥ�ɷ֢ڣ����루CH2O���������������������� ��
��2��������������NaCl������������С��粻���˷Ѵ������������ټ��� ���������� �����ϱ����������ھ��ּ�����Ӧ�����ӵijɷ��� ��
��3������Ӫ���ɷֹ��� �ࣻ�ϱ��и��ɷ�����ȷ����ԭ���� �������ۺ������������ڸ��ֳɷֶ��ܻ����װǰ��Ҫ���е��� ��������
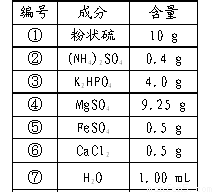
��8�֣��±���ij�����������ɷ֣���ݴ˻ش�
| ��� | �� | �� | �� | �� | �� | �� | �� |
| �ɷ� | ��״�� | ��NH4��2SO4 | K2HPO4 | MgSO4 | FeSO4 | CaCl2 | H2O |
| ���� | 10g | 0.4g | 4.0g | 9.25g | 0.5g | 0.5g | 1000ml |
��2��������������NaCl�����������С��粻���˷Ѵ������������ټ���ߣߣߣߣߣߣߣߣߣߣ����������ߣߣߣߣߣߣߣߣߣߡ�
��3������ȥ�ɷ֢ڣ����루CH2O�����������������������ߣߣߣߣߣߣߣߣߣߡ�
��4������Ӫ���ɷֹ��Уߣߣߣߣ��ࡣ
��5�����ۺ������������ڸ��ֳɷֶ��ܻ����װǰ��Ҫ���е��ǣߣߣߣߣߡ�
��6���ϱ��и��ɷ�����ȷ����ԭ���ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��7�����������������ھ��ּ�����Ӧ�����ӵijɷ��ǣߣߣߣߣߡ�
 O�����������������������ߣߣߣߣߣߣߣߣߣߡ�
O�����������������������ߣߣߣߣߣߣߣߣߣߡ�