��Ŀ����
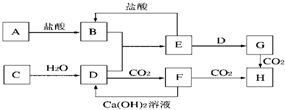
��ͼ��A��H��Ϊ��ѧ��ѧ�г��������ʣ�����֮��������ת����ϵ������A��C��Ϊ�������ʣ�C��ˮ��Ӧ����D����������壬D��H����ɫ��Ӧ���ʻ�ɫ����ͨ��״����E�����������NaOH��������ɷ������ֽⷴӦ����Ӧ���������ɵ�ˮ��������������ȥ����
��ش��������⣺
��1��B��______��H��______�����ѧʽ��
��2��д��Eת��ΪG�����ӷ���ʽ______��
��3��A�ڳ�����Ҳ����D��Һ��Ӧ����G��д���˷�Ӧ�Ļ�ѧ����ʽ______��
��ش��������⣺
��1��B��______��H��______�����ѧʽ��
��2��д��Eת��ΪG�����ӷ���ʽ______��
��3��A�ڳ�����Ҳ����D��Һ��Ӧ����G��д���˷�Ӧ�Ļ�ѧ����ʽ______��

A��C��Ϊ�������ʣ�C��ˮ��Ӧ����D����������壬D��H����ɫ��Ӧ���ʻ�ɫ����ͨ��״����E�����������NaOH��������ɷ������ֽⷴӦ����CΪNa��DΪNaOH��EΪAl��OH��3�����ͼ�е�ת����֪��FΪNa2CO3��BΪAlCl3��GΪNaAlO2��F�������̼��Ӧ����H��HΪNaHCO3��
��1��������������֪��BΪAlCl3��HΪNaHCO3���ʴ�Ϊ��AlCl3��NaHCO3��
��2��Eת��ΪG�����ӷ���ʽΪAl��OH��3+OH-�TAlO2-+2H2O���ʴ�Ϊ��Al��OH��3+OH-�TAlO2-+2H2O��
��3��A�ڳ�������D��Һ��Ӧ����G�Ļ�ѧ��ӦΪ2Al+2NaOH+2H2O�T2NaAlO2+3H2����
�ʴ�Ϊ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����
��1��������������֪��BΪAlCl3��HΪNaHCO3���ʴ�Ϊ��AlCl3��NaHCO3��
��2��Eת��ΪG�����ӷ���ʽΪAl��OH��3+OH-�TAlO2-+2H2O���ʴ�Ϊ��Al��OH��3+OH-�TAlO2-+2H2O��
��3��A�ڳ�������D��Һ��Ӧ����G�Ļ�ѧ��ӦΪ2Al+2NaOH+2H2O�T2NaAlO2+3H2����
�ʴ�Ϊ��2Al+2NaOH+2H2O�T2NaAlO2+3H2����

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д� Ŀ�����ϵ�д�
Ŀ�����ϵ�д�
�����Ŀ


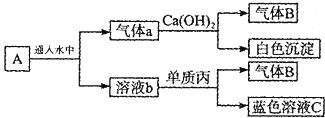
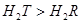
 ��Һ��Ӧ
��Һ��Ӧ