��Ŀ����
��14�֣�ʳ���к���һ������þ���������ʣ��ӵ����е����ʧ��Ҫ���������ʡ�ˮ�֡������е������Լ����ա����ȶ�����ġ���֪�������ԣ� ��Fe3����I2����ԭ�ԣ�
��Fe3����I2����ԭ�ԣ� ��I����3I2��6OH��
��I����3I2��6OH�� ��5I����3H2O�� KI��I2KI3
��5I����3H2O�� KI��I2KI3
��1��ijѧϰС��Լӵ��ν�������ʵ�飺ȡһ����ij�ӵ���(���ܺ���KIO3��KI��Mg2����Fe3��)������������ˮ�ܽ⣬����ϡ�����ữ����������Һ��Ϊ3�ݡ���һ����Һ�еμ�KSCN��Һ���Ժ�ɫ���ڶ�����Һ�м�����KI���壬��Һ�Ե���ɫ����CCl4��ȡ���²���Һ����
��ɫ����������Һ�м�������KIO3����μӵ����Լ�����Һ����ɫ��
�ټ�KSCN��Һ�Ժ�ɫ���ú�ɫ���ʵĻ�ѧʽ��_________��CCl4�����Ϻ�ɫ�����ʵĵ���ʽ��___________________��
�ڵڶ�����Һ�м�������KI�����Ӧ�����ӷ���ʽΪ_________________________��
______________________________________��
��2��KI��Ϊ�ӵ����ʳ���ڱ�������У����ڿ��������������ã�������������ʧ��
д����ʪ������KI��������Ӧ�Ļ�ѧ����ʽ��_____________________________��
��3������KI��Ϊ�ӵ����ʳ���ڱ�������У�Ϊ����ӵ��Σ�����KI�����ȶ��ԣ��ɼ��ȶ������ٵ����ʧ�������������п�����Ϊ�ȶ�������___________________��
A��Na2S2O3 B��AlCl3 C ��NaNO2
��4���Ժ�Fe2���϶��ʳ��(���費��Fe3��)����ѡ��KI��Ϊ�ӵ���������ʵ�鷽������üӵ����е�Fe2+
��14�֣�ÿ��2�֣�
��1����Fe(SCN)3
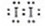 ��IO3����5I����6H��=3I2��3H2O
2Fe3����2I��=2Fe2����I2
��IO3����5I����6H��=3I2��3H2O
2Fe3����2I��=2Fe2����I2
��2��O2��4KI��2H2O=2I2��4KOH
��3��A
��4��ȡ�����üӵ�����������ˮ�У��μ�KSCN��Һ������ɫ���ٵμ��������������磺��ˮ����������ȣ�������Ѫ��ɫ����üӵ����д���Fe2+��
����������

 53������ϵ�д�
53������ϵ�д� KI3
KI3
 KI3
KI3

 ����������Ϊʳ�μӵ���Ƿ���ʣ�
����������Ϊʳ�μӵ���Ƿ���ʣ�