��Ŀ����
16��
��1������������������ġ���ɫ��Դ�����ø�ѹ������������������ȼ�ϵ�����������ܵ�һ����Ҫ��ʽ����д������ȼ�ϵ�أ��������ҺΪKOH��Һ���ĵ缫��Ӧʽ��
����O2+4e-+2H2O�T4OH-��
����2H2-4e-+4OH-�T4H2O��
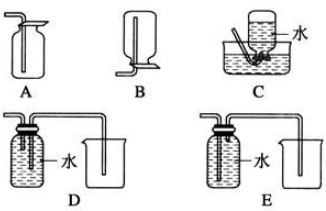
��2�����ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�ã���ͼ��ʾһ�����أ�����aΪ�������Һ��X��Y������缫�壬ͨ��������ֱ����Դ��������ش��������⣺
��Y�ĵ缫��������������д��������������������
����X��Y���Ƕ��Ե缫��a�DZ���ʳ��ˮ��ʵ�鿪ʼʱ��ͬʱ�����߸����뼸�η�̪��Һ��һ��ʱ�����X�������۲쵽����������Һ����ɫ��Ϊ��ɫ����μ���Y���ϵIJ��ʪ��ĵ��۵⻯����ֽ���۲��Ƿ���������Լ�������
��3����X��Y���Ƕ��Ե缫��a��CuSO4��Һ�����һ��ʱ��������ϲ�����������Ϊ4.48L����״���£�������������������������Ϊ25.6g��
��4����Ҫ�ø�װ�õ�⾫����ͭ�����Һaѡ��CuSO4��Һ����X�缫�IJ�����
��ͭ��Y�缫�IJ����Ǵ�ͭ��
��5����Ҫ�õ�Ʒ������������һ���������Ӧ��ѡ��ķ�����A��
| ���� | X | Y | a��Һ |
| A | �� | �� | AgNO3 |
| B | �� | �� | Fe��NO3��3 |
| C | �� | �� | AgNO3 |
| D | �� | ʯī | AgNO3 |
���� ��1������ȼ�ϼ��Ե���У�������ͨ��ȼ�ϣ�ȼ��ʧ���Ӻ����������ӷ�Ӧ����ˮ��������ͨ�������������õ��Ӻ�ˮ��Ӧ�������������ӣ�
��2����X��Y���Ƕ��Ե缫��a�DZ���NaCl��Һ��Y�缫�������ӷŵ�����������X�缫�������ӷŵ磬ͬʱ�õ缫�����������������ӣ�
��3����X��Y���Ƕ��Ե缫��a��CuSO4��Һ������������������ʧ������������������ͭ���ӵõ���������ͭ�����ݵ缫��Ӧ�͵����غ����õ���
��4����⾫����ͭ����������ͭ�����������Һaѡ��CuSO4��Һ��
��5�����ݵ��ԭ�������жϣ��Ʋ���������������ƽ������������������Һ�к��Ʋ����ӣ�
��� �⣺��1������ȼ�ϼ��Ե���У�����������ʧ���ӷ���������Ӧ���缫��ӦʽΪ2H2-4e-+4OH-=4H2O�������������õ��Ӻ�ˮ�������������ӣ���缫��ӦʽΪO2+2H2O+4e-=4OH-��
�ʴ�Ϊ��O2+4e-+2H2O�T4OH-��2H2-4e-+4OH-�T4H2O��
��2����Y���ӵ�Դ��������Ϊ���ص��������ʴ�Ϊ��������
����X��Y���Ƕ��Ե缫��a�DZ���NaCl��Һ��Y�缫�������ӷŵ������������缫��ӦʽΪ2Cl--2e-=Cl2��������ʪ��ĵ��۵⻯����ֽ���飬�۲��Ƿ������
�͵�Դ�ĸ��������ĵ缫X�����������õ缫�������ӷ����õ��ӵĻ�ԭ��Ӧ����2H++2e-=H2�������Ըõ缫����������Ũ����������ǿ�����뼸�η�̪��Һ���죬
�ʴ�Ϊ����Һ����ɫ��Ϊ��ɫ��ʪ��ĵ��۵⻯����ֽ���۲��Ƿ������
��3����X��Y���Ƕ��Ե缫��a��CuSO4��Һ������������������ʧ������������������ͭ���ӵõ���������ͭ�����ݵ缫��Ӧ�͵����غ����õ���
�����缫��Ӧ4OH--4e-=2H2O+O2���������缫��ӦCu2++2e-=Cu�����ݵ����غ�õ�2Cu��O2��4e-�������ϲ��������������״�������Ϊ4.48L�����ʵ���Ϊ$\frac{4.48L}{22.4/mol}$=0.2mol������������������ͭ�����ʵ���Ϊ0.4mol������=0.4mol��64g/mol=25.6g��
�ʴ�Ϊ��25.6��
��4����⾫����ͭ����������ͭ�����������Һaѡ��CuSO4��Һ��XΪ���������Ǿ�ͭ��Y�缫�������������Ǵ�ͭ���ʴ�Ϊ����ͭ����ͭ��
��5��Ҫ�õ�Ʒ������������һ�������������ӦΪ��������Ϊ�������������ҺΪ��������ֻ��A���ϣ��ʴ�Ϊ��A��
���� �����ۺϿ������ԭ���ķ���Ӧ�ã�Ϊ��Ƶ���㣬������ѧ���ķ��������ͼ��������Ŀ��飬ע����յ���ʵĹ���ԭ���Լ����ӵķŵ�˳��Ϊ��������Ŀ�Ĺؼ������յ�����ķ����жϣ��Ѷ��еȣ�

| A�� | ��ϩ������8���Ҽ���1���м� | |
| B�� | ��ϩ������3��̼ԭ�Ӷ���sp3�ӻ� | |
| C�� | ��ϩ������ֻ���ڼ��Լ� | |
| D�� | ��ϩ������3��̼ԭ����ͬһֱ���� |
| A�� | �������������С�ڷ�Ӧ��������� | |
| B�� | �Ͽ���ѧ�������յ�����С���γɻ�ѧ�����ų������� | |
| C�� | �÷�Ӧ��������ͼ���кͷ�Ӧ | |
| D�� | �÷�Ӧ��������ʽ������ת�����˻�ѧ�� |
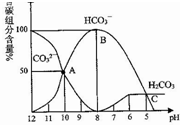 �����£�ijNa2CO3��Һ�к���̼��ֺ��������ʵ�������������Һ��pH���Ͷ������仯��ͼ������˵����ȷ���ǣ�������
�����£�ijNa2CO3��Һ�к���̼��ֺ��������ʵ�������������Һ��pH���Ͷ������仯��ͼ������˵����ȷ���ǣ�������| A�� | �ڵ��з�̪��Na2CO3��Һ����μ���������Һǡ�ñ�Ϊ��ɫ���õ���Һ | |
| B�� | ��CO2����ų��ķ�Χ��pH��5 | |
| C�� | HCO3-�ĵ���ƽ�ⳣ��Ϊ1��10-4 | |
| D�� | pH=11��Na2CO3��Һϡ�͵����Ϊԭ����100����pH=9 |
| A�� | CH4 | B�� | NH3 | C�� | H2O | D�� | CO2 |
| A�� | �٢ݢ� | B�� | �ڢܢ� | C�� | �ڢݢ� | D�� | �ۢܢ� |
| ��Ӧʱ��/min | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
| �ձ���ҩƷ��������/g | 210.0 | 206.8 | 205.9 | 205.7 | 205.6 | 205.6 | 205.6 |
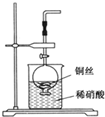
��1����Ӧ����ʱ���ռ���CO2���������״���£���
��2������Ʒ��NaHCO3������������
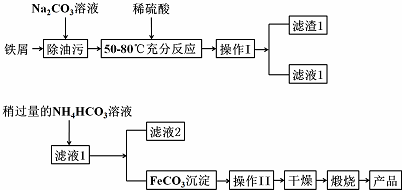
 NO���������������Ѹ�ٷ�����Ӧ����ѧ�ҷ������������в��ϵز���NO������ϸ���䴫����Ϣ��NO��������Ѫ��ϵͳ������ϵͳ�Լ��������Χ��ϵͳ�ĵ��أ�
NO���������������Ѹ�ٷ�����Ӧ����ѧ�ҷ������������в��ϵز���NO������ϸ���䴫����Ϣ��NO��������Ѫ��ϵͳ������ϵͳ�Լ��������Χ��ϵͳ�ĵ��أ�