��Ŀ����
��֪X��Y��Z��QΪ�����ڷǽ���Ԫ�أ�R�dz�����Ԫ�أ�Xԭ�ӵĵ���ռ��2�����Ӳ���ԭ���гɶԵ�������δ�ɶԵ�������2����Y�Ļ�̬ԭ����7�ֲ�ͬ�˶�״̬�ĵ��ӣ�ZԪ���ڵؿ��к�����ࣻQ�ǵ縺������Ԫ�أ�R+����ֻ���������Ӳ�����ȫ�������ӡ�
�ش��������⣺������ʱ��X��Y��Z��Q��R������Ӧ��Ԫ�ط��ű�ʾ��
��1��XԪ��Ϊ ��X��Y��Z�е�һ������������ ��
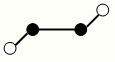
��2����֪Y2Q2���Ӵ�����ͼ��ʾ�����ֽṹ�����ģ�ͣ����߲�һ��������������

�÷�����Yԭ�ӵ��ӻ���ʽ�� ��
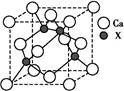
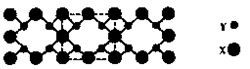
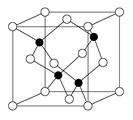
��3��X��YԪ�ؿ����γ�һ�ֳ�Ӳ�²��ϣ��侧�岿�ֽṹ��ͼ��ʾ���йظþ����˵����ȷ���� ������ȷ�𰸱�ţ���
��4����һ��AB�ͷ�����Y���ʷ��ӻ�Ϊ�ȵ����壬����һ�ֳ��õĻ�ԭ�����仯ѧʽΪ ��
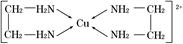
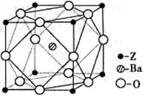
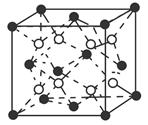
��5��R�Ļ�̬ԭ�ӵĵ����Ų�ʽΪ ��R��Z�γɵ�ij���Ӿ���ľ����ṹ��ͼ����þ���Ļ�ѧʽΪ ���þ�����ܶ�Ϊa g��cm��3����������� cm3��
�ش��������⣺������ʱ��X��Y��Z��Q��R������Ӧ��Ԫ�ط��ű�ʾ��
��1��XԪ��Ϊ ��X��Y��Z�е�һ������������ ��
��2����֪Y2Q2���Ӵ�����ͼ��ʾ�����ֽṹ�����ģ�ͣ����߲�һ��������������


�÷�����Yԭ�ӵ��ӻ���ʽ�� ��
��3��X��YԪ�ؿ����γ�һ�ֳ�Ӳ�²��ϣ��侧�岿�ֽṹ��ͼ��ʾ���йظþ����˵����ȷ���� ������ȷ�𰸱�ţ���

| A���þ������ڷ��Ӿ��� |
| B���˾����Ӳ�ȱȽ��ʯ���� |
| C������Ļ�ѧʽ��X3Y4 |
| D�������ۻ�ʱ�ƻ����ۼ� |
��5��R�Ļ�̬ԭ�ӵĵ����Ų�ʽΪ ��R��Z�γɵ�ij���Ӿ���ľ����ṹ��ͼ����þ���Ļ�ѧʽΪ ���þ�����ܶ�Ϊa g��cm��3����������� cm3��

��1��C��1�֣� N��1�֣�
��2��sp2��2�֣�
��3��bcd��3�֣�
��4��CO��2�֣�
��5��1s22s22p63s23p63d104s1��[Ar]3d104s1��2�֣� Cu2O��2�֣�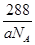 ��2�֣�
��2�֣�
��2��sp2��2�֣�
��3��bcd��3�֣�
��4��CO��2�֣�
��5��1s22s22p63s23p63d104s1��[Ar]3d104s1��2�֣� Cu2O��2�֣�
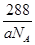 ��2�֣�
��2�֣������������1��X��Y��Z��QΪ�����ڷǽ���Ԫ�أ�R�dz�����Ԫ�أ�Xԭ�ӵĵ���ռ��2�����Ӳ���ԭ���гɶԵ�������δ�ɶԵ�������2������Ϊ�ǽ�������XΪCԪ�أ�Y�Ļ�̬ԭ����7�ֲ�ͬ�˶�״̬�ĵ��ӣ���YΪNԪ�أ�ZԪ���ڵؿ��к�����࣬��ZΪOԪ�أ�Q�ǵ縺������Ԫ�أ���QΪFԪ�أ�R������ֻ���������Ӳ�����ȫ�������ӣ���RΪCuԪ�أ��ɷ�����֪��XΪCԪ�أ�YΪNԪ�أ�ZΪOԪ�أ�QΪFԪ�أ�RΪCuԪ�أ�
��2���ӻ������=�Ҽ���+�¶Ե��Ӷ������÷�����Nԭ�ӵ��ӻ������=2+1=3������Nԭ���ӻ���ʽ��sp2�ӻ����ʴ�Ϊ��sp2�ӻ���
��3��A��X��YԪ�ؿ����γ�һ�ֳ�Ӳ�²��ϣ��ɾ��岿�ֽṹ��֪���þ���Ϊԭ�Ӿ��壬��A����
B��C-N��������C-C�����̣�C-N�����ܴ����Ծ����Ӳ�ȱȽ��ʯ����B��ȷ��
C���ṹ��ÿ��Xԭ����Χ��4��Yԭ�ӣ�ÿ��Yԭ����Χ��3��Xԭ�ӣ����Ըþ��廯ѧʽΪX3Y4����ΪC3N4����C��ȷ��
D���þ���Ϊԭ�Ӿ��壬�����ۻ�ʱ���ۼ����ƻ���û�п˷����»������������D��ȷ��
�ʴ�Ϊ��BCD��
��4�����ݶ��壬ԭ������ͬ������������ͬ�ķ��ӣ�����Ϊ�ȵ����壬������N2���ӻ�Ϊ�ȵ�����ΪCO��
��5��RΪCuԪ�أ���̬ԭ�ӵĵ����Ų�ʽΪ1s22s22p63s23p63d104s1��[Ar]3d104s1��R��Z�γɵ�ij���Ӿ���ľ�����ͭԭ�ӵĸ���Ϊ����4��1=4����ԭ�Ӹ���Ϊ����8��
 +����1��1��2�����Ըþ���Ļ�ѧʽΪCu2O���þ�����ܶ�Ϊa g��cm��3���ѣ�
+����1��1��2�����Ըþ���Ļ�ѧʽΪCu2O���þ�����ܶ�Ϊa g��cm��3���ѣ� �����������a��
�����������a�� ��V��
��V��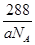 ��
��
��ϰ��ϵ�д�
�����Ŀ
 2CuCl����4H����SO42-
2CuCl����4H����SO42-