��Ŀ����
һ��������Һ̬������XY2���ڱ�״���µ�һ��������O2��ǡ����ȫȼ�գ���Ӧ����ʽΪ��XY2(l)��3O2(g)===XO2(g)��2YO2(g)����ȴ���ڱ�״���²��������������672 mL���ܶ���2.56 g/L����
(1)��ӦǰO2�������________________��
(2)������XY2��Ħ��������________________��
(3)��XY2������X��Y��Ԫ�ص���������3��16����X��Y��Ԫ�طֱ�Ϊ________��__________(дԪ�ط���)����д�� Y�����ӽṹʾ��ͼΪ ��
(4)��֪XԪ����aX��bX��cX����ԭ�ӣ�YԪ����eY��f Y ����ԭ�ӣ��������ܹ��γ� ��XY2���ӡ�
(1)672mL�� (2)76g/mol�� (3) C��S (дԪ�ط���)�� �� (4)9
�� (4)9
����

��ϰ��ϵ�д�
�����Ŀ
 �ڱ�״���µ�һ��������
�ڱ�״���µ�һ�������� ��ǡ����ȫȼ��,��Ӧ����ʽΪ:
��ǡ����ȫȼ��,��Ӧ����ʽΪ: 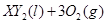 ===
===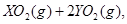 ��ȴ��,�ڱ�״���²��������������672 mL,�ܶ���2.56
��ȴ��,�ڱ�״���²��������������672 mL,�ܶ���2.56  ��:
��: 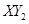 ��Ħ��������
��
��Ħ��������
��