��Ŀ����
NaCl��NaClO�����������¿ɷ�����Ӧ��ClO��+Cl��+2H+ = Cl2��+H2O��ijѧϰС�����о�����Һ(��Ҫ�ɷ�ΪNaCl��NaClO)�ı��������
��1��������Һ��NaClO�����տ����е�CO2����NaHCO3��HClO�����ʡ�д����ѧ��Ӧ����ʽ ��
��2��ȡ��������Һ�����Թ��У���������һ��Ũ�ȵ����ᣬ������ų���ͨ������װ�ü�������ijɷֿ����ж�����Һ�Ƿ���ʡ�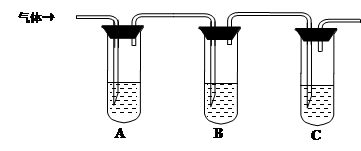
��ѡ�Լ���98%Ũ���ᡢ1%Ʒ����Һ��1.0 mol��L��1 KI-������Һ��1.0 mol��L��1NaOH������ʯ��ˮ������NaCl��Һ
���������ʵ�鷽����
| �����Լ� | Ԥ������ͽ��� |
| �Թ�A�м������� �� �Թ�B�м�1%Ʒ����Һ�� �Թ�C�мӢ� �� | ��A����Һ����ɫ��B����Һ����ɫ��C����Һ����ǡ�������Һ���ֱ��ʣ� �� ������Һδ���ʣ� �� ������Һ��ȫ���ʡ� |
��3���õζ����ⶨ����Һ��NaClO��Ũ�ȡ�ʵ�鲽�����£�
����ȡ 25.00mL����Һ������ƿ�У����������a mol��L��1 Na2SO3��Һb mL��
�ڵζ���������c mol��L��1������KMnO4��Һװ�� ������ʽ���ʽ���ζ����У�KMnO4��ʣ���Na2SO3������Ӧ������Һ����ɫ���dz��ɫ���ұ��ְ�����ں�ɫ����ʱ��ֹͣ�ζ�����¼���ݡ��ظ��ζ�ʵ��2�Σ�ƽ����������KMnO4��Һv mL��
�ζ��������漰�ķ�Ӧ�У�NaClO + Na2SO3 = NaCl+ Na2SO4 ��
2KMnO4 + 5Na2SO3+ 3H2SO4 = K2SO4 + 2MnSO4 + 5Na2SO4 + 3H2O
�ۼ��㡣����Һ��NaClO��Ũ��Ϊ mol��L��1���ú�a��b��c��v�Ĵ���ʽ��ʾ����
��16�֣�
��1��NaClO+CO2+H2O==NaHCO3+HClO ��3�֣�
��2��(��8��) �����Լ� Ԥ������ͽ��� �� 1.0mol/L KI-������Һ��2�֣�
������ʯ��ˮ��2�֣�����A����Һ����ɫ��B����Һ����ɫ(�ޱ仯)��C����Һ�������(�ޱ仯)��������Һδ���ʣ�2�֣�
����A����Һ������ɫ(�ޱ仯)��B����Һ����ɫ(�ޱ仯)��C����Һ�����������Һ��ȫ���ʣ�2�֣�
��3������ʽ ��2�֣�
�ۣ�2ab �C 5cv��/ 50 ��3�֣�
���������������1�������⣬��Ҫ��Ӧ��ΪNaClO��CO2������ΪNaHCO3��HClO��������Ԫ�ػ��ϼ۱仯�����ݹ۲취��ƽ�ɵã�NaClO+CO2+H2O==NaHCO3+HClO����2������Һ�м�������ϡ����ʱ�����ܷ����ķ�ӦΪClO��+Cl��+2H+=Cl2��+H2O��HCO3��+H+=CO2��+H2O����ų���������ܺ���������������̼����A����Һ�������ɴ��ƶ��Թ�A�м����������1.0mol/LKI��������Һ����Ϊ������KI�ܷ����û���Ӧ���������û����ĵ��ʵ������B��Ʒ����Һ����ɫ��˵��������ȫ��A����Һ���ģ���C�б���ǣ�˵��������һ���ж�����̼���壬�Թ�C�м�����������ʯ��ˮ����Ϊ������̼������������Һ��Ӧ����̼��Ƴ�����ˮ���۸�����֪����ͽ����ƶϣ���A����Һ����ɫ��B����Һ����ɫ��C����Һ������ǣ�������Һδ���ʣ�����A����Һ������ɫ��B����Һ����ɫ��C����Һ����ǣ�������Һ��ȫ���ʣ���3���٢����Ը��������Һ�����ԣ����Ӧ��װ����ʽ�ζ����У������⣬����n=c?V�������������ܵ����ʵ���Ϊab��10��3mol��ÿ�εζ����ĵĸ������Ϊ cv��10��3mol������2KMnO4+5Na2SO3+3H2SO4=K2SO4+2MnSO4+5Na2SO4+3H2O�и����ʵ�ϵ��֮�ȵ������ʵ���֮�ȣ����������������������Ϊ5cv/2��10��3mol��������������������������Ϊ��ab��10��3��5cv/2��10��3��mol������NaClO+Na2SO3=NaCl+Na2SO4�и����ʵ�ϵ��֮�ȵ������ʵ���֮�ȣ����������Ϊ��ab��10��3��5cv/2��10��3��mol������c=n/V��V=0.025L����������Ƶ����ʵ���Ũ��Ϊ��ab��10��3��5cv/2��10��3��mol��0.025L ="(2ab" �C 5cv)/ 50mol/L��
���㣺���黯ѧʵ�鷽������ƺͲⶨ��Ʒ���ȵĻ�ѧ���㣬�漰����������Һ�������̼�ķ�Ӧԭ�������ʵ�鷽��̽������������Һ�Ƿ���ʡ��ζ��ܵ�ѡ�ⶨ����������Һ�����ʵ���Ũ�ȡ����ʵ����ڻ�ѧ����ʽ�е�Ӧ�õȡ�

Ϊ̽��Cl2��Ư�۵��Ʊ����й����ʣ�ij��ȤС����Ʋ�����������ʵ��̽������ش��������⣺
��1��ʵ������������װ���Ʊ����﴿�����������밴������������������ķ��������������ӣ�H��_______��_______��_______��_______��_______�����й��ƿ���е��Լ�Ϊ_______��

��2��д����ҵ����������ʯ������ȡƯ�۵Ļ�ѧ��Ӧ����ʽ_______��
��3��ʵ������һƿ�ܷⲻ�ϵ�Ư����Ʒ�����п϶�����CaCl2�������ʵ�飬̽������Ʒ�г�CaCl2����е������������ʡ�
������������衣
����1����Ư��δ���ʣ�������Ca(ClO)2
����2����Ư��ȫ�����ʣ�������______��
����3����Ư�۲��ֱ��ʣ�������Ca(ClO)2��CaCO3��
�����ʵ�鷽��������ʵ�顣�����±���д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
��ѡ�õ�������ҩƷ���Թܡ��ιܡ������ܵĵ�����������ˮ������ˮ��Ʒ����Һ��1 mol��L-1 HCl��Һ�����Ƴ���ʯ��ˮ������ʾ�����ؼ���Ca2+��Cl-����
| | ʵ�鲽�� | Ԥ����������� |
| ����1 | ȡ��������Ư�����Թ��У��ȼ��� �ܽ���ٰ����ɵ�����ͨ�� �� | �� �������1������ �� �������2�����3������ |
| ����2 | ��ȷ��Ư�۱��ʣ�����ȡ��������Ư�����Թ��У��ȼ�������1 mol��L-1 HCl��Һ���ټ��� �� | �� �������2������ �� �������3������ |
ijʵ��С�����ͨп�̷ϸɵ���ڵĺ�ɫ�������̽����������·�����
��֪��I����ͨп�̵�صĺ�ɫ������Ҫ�ɷ�ΪMnO2��NH4Cl��ZnCl2�����ʡ�
II��������пΪ��ɫ��ĩ��������ˮ�������ᡢǿ����Һ�Ͱ�ˮ��
��ش��������⣺
��1���ڲ�����������___________��
��2��ijͬѧ������ҺA�ijɷֺ���NH4Cl��ZnCl2���������һ��ʵ�鷽������֤�������ȷ��Ҫ���ڴ���ϰ��±���ʽд��ʵ�������Ԥ������ͽ��ۡ�
��ѡ�Լ�������ˮ��2moL��L��1 HCI ��2 moL��L��1 HNO3 ��2 moL��L��1 NH3��H2O��6 moL��L��1 NaOH��0.1 moL��L��1 KSCN��0.1 moL��L��1 BaCl2��0.1 moL��L��1 AgNO3����ɫʯ����Һ����ɫʯ����ֽ
| ʵ����� | Ԥ������ | ���� |
| ����1����ȡ������ҺA��װa��b��c��֧�Թܣ���a�Թܣ�__ __________________________ | �а�ɫ�������� | ˵����ҺA����Cl�� |
| ����2����b�Թܣ�__________ __________________________ | ______________________ | _______________________ |
| ����3����c�Թܣ�__________ __________________________ | �Ȳ���_______________, ��____________________ | ˵����ҺA����Zn2+ |
��3��ȡ��������c�����Թܣ��μ���˫��ˮ���۲쵽�����������д���÷�Ӧ�Ļ�ѧ����ʽ��_______________��
��4��Ϊ�ⶨ�ϸɵ���ж������̵�������������������ʵ�飺ȷ��ȡag��ǧ��ع��壬����ϡ���ᣬ����⻯����Һ����ַ�Ӧ����bmol/L��������Ʊ���Һ�ζ����õ�����ָʾ�����ζ����յ㣬�ظ�ʵ�飬ƽ��������������Ʊ���Һ�����ΪvmL����ϵ���ж������̵����������ļ������ʽΪ��________________________________��
���ζ��йط�Ӧ��MnO2+2I��+4H+=Mn2++I2+2H2O��I2+2S2O32��=2I��+S4O62����
���ͷ���һ����������Ʒ����ʳƷ�Ļ�ѧ���ɼ�����С�մ���(̼�����)�������е�����������ɡ�ij�о���ѧϰС��Ϊ̽����ͬƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣���������ʵ�顣
��������衿
��1������1����С�մ�ͳ�����ɣ�
����2����С�մ��������ɣ�
����3����________________��ɡ�
�����������̡�
Ϊ̽��ijƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣�ijͬѧ�������ʵ�飬�õ���������
��2���÷��ͷ۵ijɷ�Ϊ________ (�ѧʽ����
��3����һƷ�Ƶķ��ͷ۵Ļ�ѧ��ɿ���Ϊ����2������������ʵ����֤��д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
| ʵ�鲽�� |  Ԥ��������� Ԥ��������� |
| 1��ȡ������Ʒ����ϡ�������Һ�ֳ����� | |
| 2�� _______________________________________ | |
| 3�� ________________________________________ | |
��4����һƷ�Ƶķ��ͷ۵Ļ�ѧ���ΪС�մ��̼����泥�Ϊ̽���仯ѧʽ[��ѧʽ�ɱ�ʾΪnNaHCO3��m NH4HCO3]����ȡ4.05 g�ò�Ʒ���ձ����ܽ������________���100 mL��Һ������Һ��ȡ25.00mL��Һ���μ��������ᣬ���ɵ�����ͨ���������ʯ��ˮ�У����ɵİ�ɫ�������������Ϊ1.25 g����÷��ͷ۵Ļ�ѧʽΪ ��
����Է���������NaHCO3��84 NH4HCO3��79 CaCO3��100��
����ʵ���ܴﵽ��ӦĿ�ĵ���
| A����ͼ��װ����ȡ���ռ����� | B����ͼ��װ����ȡ���ռ���ϩ |
| C����ͼ��װ�ý��������ճɻ� | D����ͼ��װ����ȡ�������� |
����ҩƷ��װ�ú������������Ӧʵ����ǣ� ��

| A����Ȫʵ�� |
| B��ʵ������ȡ���ռ����� |
| C���Ʊ����������� |
| D����֤�����Ƿ���̼̼˫�� |



