��Ŀ����
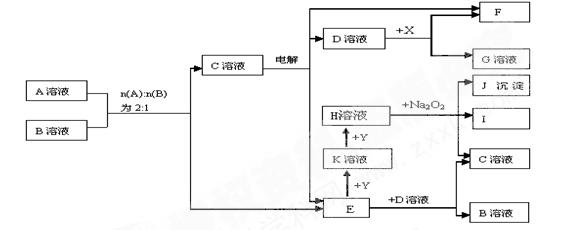
��8�֣�ͼ��A��K������ѧ��ѧ�г������ʡ�A��һ���ᣬE��F��Iͨ��״���������壬����E�д̼�����ζ��X��Y�dz����Ľ������ʣ�JΪ���ɫ�������μӷ�Ӧ��ˮ�Լ����ɵ�ˮ��ͼ�о�����ȥ��
��ش��������⣺
��1��E�ĵ���ʽΪ _____________�� J�Ļ�ѧʽΪ ��
��2��д������Һ��ٷ�Ӧ�����ӷ���ʽ���������������������������� ��
��3��д��D��Һ��ط�Ӧ�����ӷ���ʽ __________��
��4��д��A��B �� C��E�Ļ�ѧ����ʽ ______ __��

��ش��������⣺
��1��E�ĵ���ʽΪ _____________�� J�Ļ�ѧʽΪ ��
��2��д������Һ��ٷ�Ӧ�����ӷ���ʽ���������������������������� ��
��3��д��D��Һ��ط�Ӧ�����ӷ���ʽ __________��
��4��д��A��B �� C��E�Ļ�ѧ����ʽ ______ __��
����8�֣�
��1��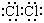 ����1�֣�Fe(OH)3����1�֣�
����1�֣�Fe(OH)3����1�֣�
��2��2Fe3+ + Fe = 3Fe2+��2�֣�
��3��2Al + 2H2O + 2OH- = 2AlO2- + 3H2����2�֣�
��4��2HCl + NaClO =" NaCl" + H2O + Cl2����2�֣�
��1��
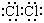 ����1�֣�Fe(OH)3����1�֣�
����1�֣�Fe(OH)3����1�֣� ��2��2Fe3+ + Fe = 3Fe2+��2�֣�
��3��2Al + 2H2O + 2OH- = 2AlO2- + 3H2����2�֣�
��4��2HCl + NaClO =" NaCl" + H2O + Cl2����2�֣�
��

��ϰ��ϵ�д�
�����Ŀ
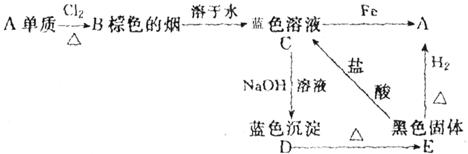
 B��__
B��__ _______��C��_________��D��_________��E��_________��
_______��C��_________��D��_________��E��_________��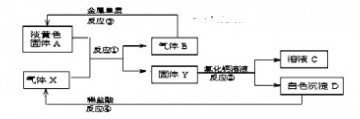

 C�е���������Ҫ��_
C�е���������Ҫ��_