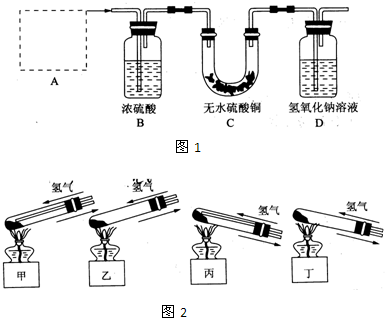
��Ŀ����
��8�֣�10��ʱ����NaHCO3������Һ����ø���Һ��pH�������±仯��
| �¶ȣ��棩 | 10 | 20 | 30 | ������к���ȴ��50�� |
| pH | 8.3 | 8.4 | 8.5 | 8.8 |
 ��
����ͬѧ��Ϊ����ҺpH���ߵ�ԭ����NaHCO3���ȷֽ⣬������Na2CO3�����ƶ�Na2CO3��ˮ��̶� ������ڡ���С�ڡ���NaHCO3��
��ͬѧ��Ϊ�ס��ҵ��ж϶�����֡�����Ϊ��
��1��ֻҪ�ڼ�����е���Һ�м����������Լ�X��������������
 �����ж���ȷ���Լ�X�� ��A��Ba(OH)2��Һ B��NaOH��Һ C��BaCl2��Һ D������ʯ��ˮ
�����ж���ȷ���Լ�X�� ��A��Ba(OH)2��Һ B��NaOH��Һ C��BaCl2��Һ D������ʯ��ˮ��2�������Ⱥ����Һ��ȴ��10�棬����Һ��pH (����ڡ��������ڡ����ڡ���8.3������ж���ȷ��
��8�֣�
HCO3��+H2O  H2CO3+OH��(2��)�� ����(2��) ����C (2��)��
H2CO3+OH��(2��)�� ����(2��) ����C (2��)�� �Ƶ���(2��)
�Ƶ���(2��)
����

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ