��Ŀ����
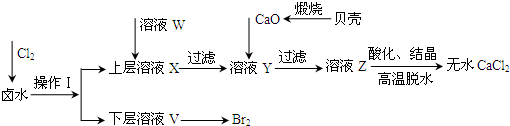
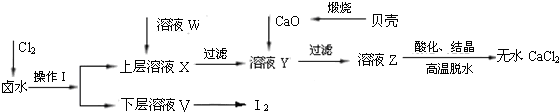
ʵ�����Ժ���Ca2+��Mg2+��Cl-��SO42-��I-�����ӵ�±ˮΪ��Ҫԭ�ϣ��Ʊ���ˮCaCl2��I2���������£�����֪Cl2��I-�ɷ�����Ӧ��Cl2+2I-=2Cl-+I2��
��1��������ʹ�õ��Լ���
��2��������ҺW��Ŀ����
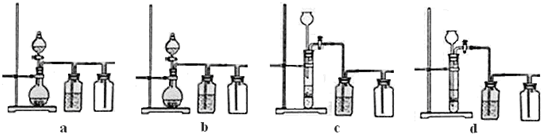
��3��ʵ�����ñ�����ϡ���ᷴӦ�Ʊ����ռ�CO2���壬����װ���к�������

��4����ƺ���ʵ����֤H2SO3����ǿ��H2CO3��������ѡ��
��ѡ���Լ���CO2��SO2��Na2CO3��Һ��NaHCO3��Һ��NaHSO3��Һ������ˮ������ʯ��ˮ������KMnO4 ��Һ��Ʒ����Һ��pH��ֽ��
��дʵ�鱨�棺

��1��������ʹ�õ��Լ���
CCl4
CCl4
��������Ҫ��������������Һ©��
��Һ©��
����2��������ҺW��Ŀ����
��ȥ��Һ�е�SO42-
��ȥ��Һ�е�SO42-
����CaO������ҺY��pH�����Գ�ȥMg2+���ữ��ҺZʱʹ�õ��Լ�������
����
����3��ʵ�����ñ�����ϡ���ᷴӦ�Ʊ����ռ�CO2���壬����װ���к�������
b��d
b��d
��
��4����ƺ���ʵ����֤H2SO3����ǿ��H2CO3��������ѡ��
��ѡ���Լ���CO2��SO2��Na2CO3��Һ��NaHCO3��Һ��NaHSO3��Һ������ˮ������ʯ��ˮ������KMnO4 ��Һ��Ʒ����Һ��pH��ֽ��
��дʵ�鱨�棺
| ʵ�鲽�� | ���� | ���� |
��SO2����ͨ�� NaHCO3��Na2CO3��Һ�� ����KMnO4��Һ�� Ʒ����Һ�� ����ʯ��ˮ ��SO2����ͨ�� NaHCO3��Na2CO3��Һ�� ����KMnO4��Һ�� Ʒ����Һ�� ����ʯ��ˮ |
NaHCO3��Na2CO3��Һ�����ݣ� Ʒ����Һ����ɫ�� ����ʯ��ˮ����� NaHCO3��Na2CO3��Һ�����ݣ� Ʒ����Һ����ɫ�� ����ʯ��ˮ����� |
H2SO3����ǿ��H2CO3 |
��������1�����ʵ⼫�������л��ܼ������Կ��Բ�����ȡ�ķ�������ȡ����Ҫ�����Ƿ�Һ©����
��2��ʵ�����Ժ���Ca2+��Mg2+��Cl-��SO42-��I-�����ӵ�±ˮΪ��Ҫԭ�ϣ��Ʊ���ˮCaCl2��I2��Ӧ��������е�Mg2+��SO42-�������ӳ�ȥ���������̿��Կ����ֱ�ͨ����������Һ�е�I-�����ⵥ�ʣ������Ȼ�����ȥSO42-����CaO������ҺY��pH�����Գ�ȥMg2+��
��3��CO2���ܶȴ��ڿ����еģ����Բ��������ſ������ռ�CO2���壮�����ó���©��ʱ������©�����¶˱�����뵽��Һ�У��Է�ֹCO2����ӳ���©���лӷ�������
��4��H2SO3��H2CO3�����ڶ�Ԫ�ᣬҪ��������ǿ�������Բ��ö��ַ�������������ˮ��ԭ������Խ����Ӧ��ǿ���ξ�Խ��ˮ�⣩��Ҳ�������ý�ǿ������ȡ�������������飮
��2��ʵ�����Ժ���Ca2+��Mg2+��Cl-��SO42-��I-�����ӵ�±ˮΪ��Ҫԭ�ϣ��Ʊ���ˮCaCl2��I2��Ӧ��������е�Mg2+��SO42-�������ӳ�ȥ���������̿��Կ����ֱ�ͨ����������Һ�е�I-�����ⵥ�ʣ������Ȼ�����ȥSO42-����CaO������ҺY��pH�����Գ�ȥMg2+��
��3��CO2���ܶȴ��ڿ����еģ����Բ��������ſ������ռ�CO2���壮�����ó���©��ʱ������©�����¶˱�����뵽��Һ�У��Է�ֹCO2����ӳ���©���лӷ�������
��4��H2SO3��H2CO3�����ڶ�Ԫ�ᣬҪ��������ǿ�������Բ��ö��ַ�������������ˮ��ԭ������Խ����Ӧ��ǿ���ξ�Խ��ˮ�⣩��Ҳ�������ý�ǿ������ȡ�������������飮
����⣺��1����������ǿ�����ԣ�ͨ����������Խ���Һ�е�I-�����ɵⵥ�ʣ���Ϊ���ʵ⼫�������л��ܼ������Կ��Բ�����ȡ�ķ�������ȡ����Ҫ�����Ƿ�Һ©����������ͼ��֪���ʵ����²㣬��˸��л��ܼ����ܶ�Ҫ��ˮ�Ĵ��Ҳ�����ˮ�����Ը��Լ���CCl4��
�ʴ�Ϊ�����Ȼ�̼����Һ©����
��2������SO42-����Ca2+����γ����Ե�����ƶ�Ӱ���Ȼ��Ƶ��Ʊ�����˱����ȥ����Ϊ���������µ����ʣ������ữ��ҺZʱ��ʹ�õ��Լ�Ӧ��Ϊ���ᣬ
�ʴ�Ϊ����ȥ��Һ�е�SO42-�����
��3��ʵ������ȡCO2���ص��ǹ����Һ�巴Ӧ�Ҳ���Ҫ���ȣ����������ӷ���������ռ�֮ǰ��Ҫ��ȥ�ӷ���HCl���壬��ΪCO2���ܶȴ��ڿ����еģ����Բ��������ſ������ռ�CO2���壮�����ó���©��ʱ������©�����¶˱�����뵽��Һ�У��Է�ֹCO2����ӳ���©���лӷ����������ѡ��b��d��ȷ��
�ʴ�Ϊ��b��d��
��4��H2SO3��H2CO3�����ڶ�Ԫ�ᣬҪ��������ǿ�������Բ��ö��ַ�������������ˮ��ԭ������Խ����Ӧ��ǿ���ξ�Խ��ˮ�⣩��Ҳ�������ý�ǿ������ȡ�������������飬����SO2��CO2���������ʹ����ʯ��ˮ����ǣ�����CO2������ͨ�����ʯ��ˮ֮ǰ�����ȥCO2�����л��е�SO2���壬������֤SO2�Ѿ�������
������KMnO4��Һ������������Ʒ����֤������SO2����ͨ��NaHCO3��Na2CO3��Һ��NaHCO3��Na2CO3��Һ���������ɶ�����̼��������̼ʹ����ʯ��ˮ����ǣ�˵��H2SO3����ǿ��H2CO3��
�ʴ�Ϊ��
�ʴ�Ϊ�����Ȼ�̼����Һ©����
��2������SO42-����Ca2+����γ����Ե�����ƶ�Ӱ���Ȼ��Ƶ��Ʊ�����˱����ȥ����Ϊ���������µ����ʣ������ữ��ҺZʱ��ʹ�õ��Լ�Ӧ��Ϊ���ᣬ
�ʴ�Ϊ����ȥ��Һ�е�SO42-�����
��3��ʵ������ȡCO2���ص��ǹ����Һ�巴Ӧ�Ҳ���Ҫ���ȣ����������ӷ���������ռ�֮ǰ��Ҫ��ȥ�ӷ���HCl���壬��ΪCO2���ܶȴ��ڿ����еģ����Բ��������ſ������ռ�CO2���壮�����ó���©��ʱ������©�����¶˱�����뵽��Һ�У��Է�ֹCO2����ӳ���©���лӷ����������ѡ��b��d��ȷ��
�ʴ�Ϊ��b��d��
��4��H2SO3��H2CO3�����ڶ�Ԫ�ᣬҪ��������ǿ�������Բ��ö��ַ�������������ˮ��ԭ������Խ����Ӧ��ǿ���ξ�Խ��ˮ�⣩��Ҳ�������ý�ǿ������ȡ�������������飬����SO2��CO2���������ʹ����ʯ��ˮ����ǣ�����CO2������ͨ�����ʯ��ˮ֮ǰ�����ȥCO2�����л��е�SO2���壬������֤SO2�Ѿ�������
������KMnO4��Һ������������Ʒ����֤������SO2����ͨ��NaHCO3��Na2CO3��Һ��NaHCO3��Na2CO3��Һ���������ɶ�����̼��������̼ʹ����ʯ��ˮ����ǣ�˵��H2SO3����ǿ��H2CO3��
�ʴ�Ϊ��
| ʵ�鲽�� | ���� | ���� |
| ��SO2����ͨ�� NaHCO3��Na2CO3��Һ�� ����KMnO4��Һ�� Ʒ����Һ�� ����ʯ��ˮ |
NaHCO3��Na2CO3��Һ�����ݣ� Ʒ����Һ����ɫ�� ����ʯ��ˮ����� |
H2SO3����ǿ��H2CO3 |
������������Ҫ���鳣���Ļ���ʵ�������������ʹ�á����ӵij��Ӻ�ʵ�鷽����ƣ��ۺ���ǿ��ƽʱע�����֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�
�����Ŀ