��Ŀ����
6��ij�������A��������������������ܶ���42��������ױ��������к���̼̼˫�����˴Ź������ױ���������ֻ��һ�����͵��⣮��1��A�Ľṹ��ʽΪ��CH3��2C=C��CH3��2������Ϊ2��3-����-2-��ϩ��
��2��A�е�̼ԭ���Ƿ���ͬһƽ�棿�ǣ���ǡ����ǡ�����A�����Ƿ����˳���칹�����ǡ�����
��3����֪A��B��C������ת����ϵ��A$��_{��}^{Br_{2}/CCl_{4}}$B$��_{�ڡ�}^{NaOH��}$$\stackrel{C}{��{C}_{6}{H}_{10}��}$��Ӧ�ڵĻ�ѧ����ʽΪ��CH3��2CBrCBr��CH3��2+2NaOH$��_{��}^{�Ҵ�}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O����Ӧ����������ȥ��Ӧ��
���� ij�������A��������������������ܶ���42����A����Է�������Ϊ84��������ױ��������к���̼̼˫�������Ը�����Cԭ�Ӹ���=$\frac{84}{12}$=6��12������ʽΪC6H12���˴Ź������ױ���������ֻ��һ�����͵��⣬��A�ṹ��ʽΪ��CH3��2C=C��CH3��2��A���巢���ӳɷ�Ӧ����B��BΪ��CH3��2CBrCBr��CH3��2��B������ȥ��Ӧ����C��C�ṹ��ʽΪCH2=C��CH3��C��CH3��=CH2���ݴ˷������
��� �⣺ij�������A��������������������ܶ���42����A����Է�������Ϊ84��������ױ��������к���̼̼˫�������Ը�����Cԭ�Ӹ���=$\frac{84}{12}$=6��12������ʽΪC6H12���˴Ź������ױ���������ֻ��һ�����͵��⣬��A�ṹ��ʽΪ��CH3��2C=C��CH3��2��A���巢���ӳɷ�Ӧ����B��BΪ��CH3��2CBrCBr��CH3��2��B������ȥ��Ӧ����C��C�ṹ��ʽΪCH2=C��CH3��C��CH3��=CH2��
��1��A�Ľṹ��ʽΪ��CH3��2C=C��CH3��2��̼̼˫����2��C����2��3��C������1������������Ϊ��2��3-����-2-��ϩ��
�ʴ�Ϊ����CH3��2C=C��CH3��2��2��3-����-2-��ϩ��
��2��A������ϩ�ṹ������A�е�̼ԭ�Ӷ�����ͬһƽ�棻���ڣ�CH3��2C=C��CH3��2���ӣ�̼̼˫�����˺��е�ȡ������ͬ�����Բ�����˳���칹��
�ʴ�Ϊ���ǣ���
��3��BΪ��CH3��2CBrCBr��CH3��2��B������ȥ��Ӧ����C��C�ṹ��ʽΪCH2=C��CH3��C��CH3��=CH2����Ӧ�ڵĻ�ѧ����ʽΪ����CH3��2CBrCBr��CH3��2+2NaOH$��_{��}^{�Ҵ�}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O��
�ʴ�Ϊ����CH3��2CBrCBr��CH3��2+2NaOH$��_{��}^{�Ҵ�}$CH2=C��CH3��C��CH3��=CH2+2NaBr+2H2O����ȥ��Ӧ��
���� ���⿼���л����ƶϣ���Ŀ�Ѷ��еȣ��漰ϩ����±����֮���ת������ȷ��Ӧ��������Ӧ�����ǽⱾ��ؼ���ע����ȥ��Ӧ�ϼ��ͳɼ���ʽ������������ѧ���ķ������������������Ӧ�û���֪ʶ��������

| A�� | �������м���Ũ�������ַ�������˵��Ũ���������ˮ�� | |
| B�� | Ũ�����ڹ�������ɫ��ƣ�˵��Ũ����ȶ� | |
| C�� | �����£�Ũ����������������棬˵������Ũ�����Ӧ | |
| D�� | Ũ���᳣������ͭ����Ӧ������ʱ���ܷ�����Ӧ |
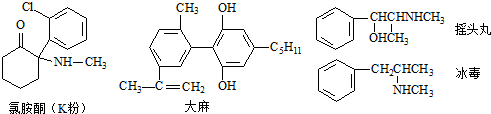
| A�� | ���ֶ�Ʒ�ж����б����������ڷ����� | |
| B�� | ҡͷ�辭��ȥ���ӳɷ�Ӧ���Ƶñ��� | |
| C�� | 1mol��������ˮ��Ӧ�������4mol Br2 | |
| D�� | �Ȱ�ͪ�����л�������̼ԭ�ӿ��ܹ�ƽ�� |
| A�� | 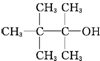 | B�� | CH3��CH2��5CH2OH | C�� | 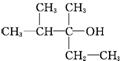 | D�� | 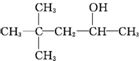 |
 ��ijһ�ݻ�Ϊ5L���ܱ������ڣ�����0.2mol��CO��0.2mol ��H2O��g�����ڴ������ڵ������¸��¼��ȣ��������·�Ӧ��CO��g��+H2O��g���TCO2��g��+H2��g������Ӧ�ų���������Ӧ��CO2��Ũ����ʱ��仯�������ͼ��ʾ��
��ijһ�ݻ�Ϊ5L���ܱ������ڣ�����0.2mol��CO��0.2mol ��H2O��g�����ڴ������ڵ������¸��¼��ȣ��������·�Ӧ��CO��g��+H2O��g���TCO2��g��+H2��g������Ӧ�ų���������Ӧ��CO2��Ũ����ʱ��仯�������ͼ��ʾ�� ��Ȼ���������������о��й㷺��Ӧ�ã�
��Ȼ���������������о��й㷺��Ӧ�ã�