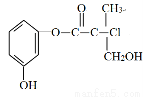
��Ŀ����
�ۺ��⣺
������������Դ��Ҳ����Ҫ�Ļ���ԭ�ϣ����������������ⷽ��������������⣺
��1������һ��H2S�ȷֽⷨ����ӦʽΪ��2H2S��g�� 2H2��g��+S2��g����H
2H2��g��+S2��g����H
�ں����ܱ������У��ⶨH2S�ֽ��ת���ʣ�H2S����ʼŨ�Ⱦ�Ϊcmol/L�����ⶨ�������ͼ��ͼ������a��ʾH2S��ƽ��ת�������¶ȹ�ϵ������b��ʾ��ͬ�¶��·�Ӧ������ͬʱ��δ�ﵽ��ѧƽ��ʱH2S��ת���ʡ�
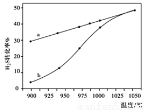
�١�H0��������������������=������
����985��ʱ����Ӧ��tmin�ﵽƽ�⣬��ʱH2S��ת����Ϊ40%����tmin�ڷ�Ӧ����v��H2��=
���ú�c��t�Ĵ���ʽ��ʾ����
����˵�����¶ȵ����ߣ�����b������a�ӽ���ԭ��
��2������������CaOΪ�����壬��������ʣ���C�ƣ���ˮ������Ӧ��ȡH2�������Ҫ��Ӧ���£�
I��C��s��+H2O��g���TCO��g��+H2��g����H=+131.0kJ/mol
��CO��g��+H2O��g���TCO2��g��+H2��g����H=-43kJ/mol
��CaO��s��+CO2��g���TCaCO3��s����H=-178.3kJ/mol
�ټ��㷴ӦC��s��+2H2O��g��+CaO��s�� CaCO3��s��+2H2��g���ġ�H=kJ/mol��
CaCO3��s��+2H2��g���ġ�H=kJ/mol��
��K1��K2��K3�ֱ�Ϊ��ӦI�����ƽ�ⳣ������ƽ�ⳣ��K=����K1��K2��K3��ʾ����
�ڶ��ڿ��淴ӦC��s��+2H2O��g��+CaO��s�� CaCO3��s��+2H2��g������ȡ���´�ʩ�������H2���ʵ��ǡ�������ĸ��
CaCO3��s��+2H2��g������ȡ���´�ʩ�������H2���ʵ��ǡ�������ĸ��
A��������ϵ���¶�
B��ѹ�����������
C������CaO����
D��ѡ���ʵ��Ĵ���
��3�������������ü״�������ˮ������ Ӧ��ȡH2����Ӧ����ʽ���£�
Ӧ��ȡH2����Ӧ����ʽ���£�
CH3OH��g��+H2O��g�� CO2��g��+3H2��g����H��+49.4kJ/mol
CO2��g��+3H2��g����H��+49.4kJ/mol
һ���¶��£����ݻ�Ϊ2L���ܱպ��������г���1molCH3OH��g����3molH2O��g�����ﵽƽ��״̬ʱ����������19.76kJ����
�ٴ�ƽ��ʱ��������ѹǿ�Ƿ�Ӧǰ�ı���
�ڴ��¶��µĸ÷�Ӧ��ƽ�ⳣ���ǣ�������λ��Ч���֣���
| A�� | �������Ȼ���ȹ����������ı仯����Ϊ��Ӧ�� | |
| B�� | �Ȼ�ѧ����ʽ�еĻ�ѧ������ֻ��ʾ���ʵ����������Ƿ��� | |
| C�� | ��Ҫ���Ȳ��ܷ����ķ�Ӧһ�������ȷ�Ӧ | |
| D�� | 1mol����ȼ��������̬ˮ�Ͷ�����̼���ų��������Ǽ���ȼ���� |
| A�� | һ��������6.4g SO2������������Ӧ����SO3��ת�Ƶ�����Ϊ0.2 NA | |
| B�� | 1 mol AlCl3������״̬ʱ��������Ϊ0.4NA | |
| C�� | ����£�11.2 L������ȫ����1 Lˮ�У�������Һ��Cl-��ClO-����������֮��ΪNA | |
| D�� | 6.4 g��S2��S4��S8��ɵĻ���ﺬ��ԭ����Ϊ0.2 NA |