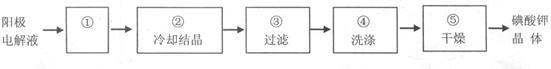
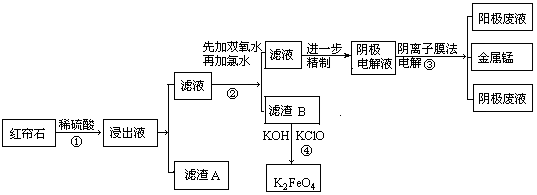
��Ŀ����
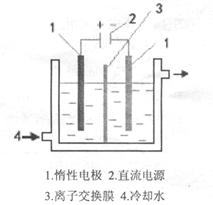
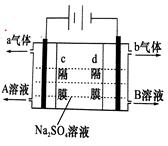
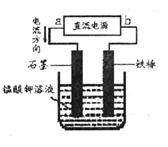
��ҵƷ����������Һ�к���ijЩ����������ʣ��������ӽ���Ĥ������ᴿ��������װ�������ӽ���Ĥ��ֻ����������ͨ�������乤��ԭ����ͼ��ʾ������˵������ȷ����
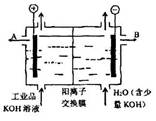

| A���������Ͽ�����Fe������������ʲ�����缫�Ϸŵ� |
| B���õ��۵�������ӦʽΪ��4OH�D�D 4e��= 2H2O+O2�� |
| C��ͨ��õ�������������ҺpH���С |
| D����ȥ���ʺ�����������Һ�ӳ���B���� |
C
����������õ��ط�Ӧ��ʵ���ǵ��ˮ��������Ӧ��2H2O+2e-=2OH-+H2��,ͨ�����������pH����������Ӧ��2H2O-4e-=4H++O2��,K+ͨ�������ӽ���Ĥ���������������OH-����KOH��Һ����ʽ��B�ڵ������ɴ˿��Եó�A��B��D��ȷ��C����ѡC��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ

 2H2SO4��4NaOH��O2����2H2��
2H2SO4��4NaOH��O2����2H2��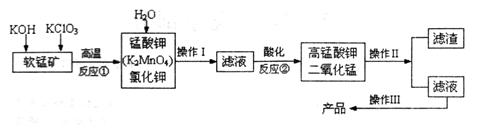

 R2Cu���л��ࣩ+ 2H����ˮ�ࣩ
R2Cu���л��ࣩ+ 2H����ˮ�ࣩ