��Ŀ����
ijͬѧ̽�����������H2O2�ֽ����ʵ�Ӱ�죬����H2O2Ũ��Ϊ5%��10%��ʵ���¶�Ϊ20�桢40�棬�����Լ���1mol/LFeCl3��Һ��ÿ��ʵ��H2O2������Ϊ2mL��FeCl3��Һ2�Σ�
��1�����������ʵ����Ʊ���
��2��ʵ��ܲ������ݵ����ʱ�ʵ��ڿ죮��������Ӧ���ʲ����ԭ����______��
��3���Ͼ�ӡˢ��·��Ļ������ÿ�ʵ����Դ��������������룬�ܵõ��ǽ�����ĩ�ͽ�����ĩ����H2O2��H2SO4�Ļ����Һ���ܳ�ӡˢ��·�������ĩ�е�ͭ����Һ�����ɫ��д��������Ӧ�Ļ�ѧ����ʽ��______��
��������������ͬ��ӡˢ��·��Ľ�����ĩ��10%H2O2��3.0mol/LH2SO4��Һ��������ò�ͬ�¶���ͭ��ƽ���ܽ����ʣ����±���
���¶ȸ���40��ʱ��ͭ��ƽ���ܽ��������ŷ�Ӧ�¶ȵ����߶��½�������Ҫԭ���ǣ�______��
��1�����������ʵ����Ʊ���
| ʵ���� | �¶�/�� | H2O2Ũ��/% | �����Լ� | ʵ��Ŀ�� |
| �� | 20 | 5 | �� | ����ʵ��ٺ͢�̽��������H2O2�ֽ����ʵ�Ӱ�죻 ����ʵ��ں͢�̽���¶ȶԸ÷�Ӧ���ʵ�Ӱ�죻 ����ʵ��ں͢�̽��H2O2Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죮 |
| �� | 20 | 5 | FeCl3��Һ | |
| �� | ______ | ______ | ______ | |
| �� | ______ | ______ | ______ |
��3���Ͼ�ӡˢ��·��Ļ������ÿ�ʵ����Դ��������������룬�ܵõ��ǽ�����ĩ�ͽ�����ĩ����H2O2��H2SO4�Ļ����Һ���ܳ�ӡˢ��·�������ĩ�е�ͭ����Һ�����ɫ��д��������Ӧ�Ļ�ѧ����ʽ��______��
��������������ͬ��ӡˢ��·��Ľ�����ĩ��10%H2O2��3.0mol/LH2SO4��Һ��������ò�ͬ�¶���ͭ��ƽ���ܽ����ʣ����±���
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| ͭ��ƽ���ܽ����� ����10-3mol/��L?min���� | 7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
��1���ۡ�ʵ��ں͢�̽���¶ȶԸ÷�Ӧ���ʵ�Ӱ�죬�ʢڢ۵��¶Ȳ�ͬ������������ȫ��ͬ��
�ܡ�ʵ��ں͢�̽��H2O2Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죬�ʢںܵ͢�H2O2Ũ�Ȳ�ͬ������������ͬ��
�ʴ�Ϊ��
��2��ʵ��ܲ������ݵ����ʱ�ʵ��ڿ죬����Ӧ���ʲ����ԭ���ǣ�����������ͬʱ��H2O2Ũ�����ֽ����ʼӿ죻
�ʴ�Ϊ������������ͬʱ��H2O2Ũ�����ֽ����ʼӿ죻
��3��ͭ��H2O2��H2SO4��Ӧ����������ͭ����Ӧ����ʽ�ǣ�Cu+H2O2+H2SO4
CuSO4+2H2O�������¶ȸ���40��������¶ȵ����ߣ�H2O2�ķֽ����ʼӿ죬ʹ��Һ��H2O2��Ũ�Ƚ��ͼӿ죬��ͭ���ܽ����ʵ�Ӱ�쳬�����¶����߶�ͭ�ܽ����ʵ�Ӱ�죬����ͭ���ܽ�������С��
�ʴ�Ϊ��Cu+H2O2+H2SO4
CuSO4+2H2O���¶ȸ���40��������¶ȵ����ߣ�H2O2�ķֽ����ʼӿ죬ʹ��Һ��H2O2��Ũ�Ƚ��ͼӿ죬��ͭ���ܽ����ʵ�Ӱ�쳬�����¶����߶�ͭ�ܽ����ʵ�Ӱ�죬����ͭ���ܽ�������С��
�ܡ�ʵ��ں͢�̽��H2O2Ũ�ȶԸ÷�Ӧ���ʵ�Ӱ�죬�ʢںܵ͢�H2O2Ũ�Ȳ�ͬ������������ͬ��
�ʴ�Ϊ��
| �� | 40 | 5 | FeCl3��Һ | ��2�֣� |
| �� | 20 | 10 | FeCl3��Һ | ��2�֣� |
�ʴ�Ϊ������������ͬʱ��H2O2Ũ�����ֽ����ʼӿ죻
��3��ͭ��H2O2��H2SO4��Ӧ����������ͭ����Ӧ����ʽ�ǣ�Cu+H2O2+H2SO4
| ||
�ʴ�Ϊ��Cu+H2O2+H2SO4
| ||

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
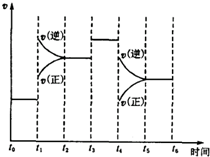
 2Z�ڷ�Ӧ�����еķ�Ӧ���ʣ�v����ʱ�䣨t���Ĺ�ϵ���ߣ�����������ȷ���ǣ�������
2Z�ڷ�Ӧ�����еķ�Ӧ���ʣ�v����ʱ�䣨t���Ĺ�ϵ���ߣ�����������ȷ���ǣ�������