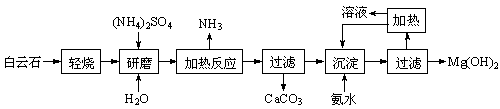
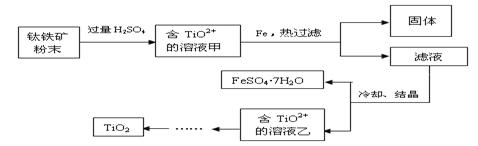
��Ŀ����
̼��﮹㷺Ӧ�����մɺ�ҽҩ��������﮻�ʯ����Ҫ�ɷ�ΪLiAlSi2O6��Ϊԭ�����Ʊ�Li2CO3�Ĺ����������£�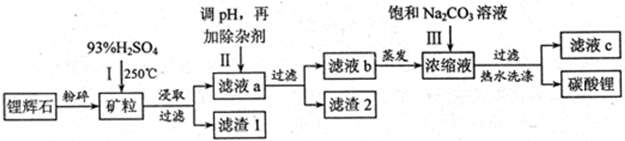
��֪����2LiAlSi2O6��H2SO4(Ũ)  Li2SO4��Al2O3��4SiO2��H2O��
Li2SO4��Al2O3��4SiO2��H2O��
��Fe3+��Al3+��Fe2+��Mg2+������������ʽ��ȫ����ʱ����Һ��PH�ֱ�Ϊ3.2��4.7��9.0��11.1
��ijЩ���ʵ��ܽ�ȣ�S�����ұ�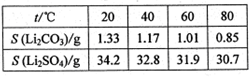
��ش��������⣺
��1��﮻�ʯ��Ũ�����ȡ֮ǰҪ�����ϸ������Ŀ���� ��
��2����Һa�к���Li+��SO42-,������Fe3+��Al3+��Fe2+��Mg2+ ��Ca2+��Na+�����ʣ���������ڽ����¼���ʯ��ʯ�Ե�����Һ��pH��6.0��6.5����ʱ���������������� ��
��3�����������Һa�м���ij��Ӽ�����Ϊ������H2O2��Һ��ʯ�����Na2CO3��Һ��������Ӧ�����ӷ���ʽ�� ��
��4��������м��뱥��Na2CO3��Һ���˺���Ҫ����ˮϴ�ӵ�ԭ���� ��
��5������Һc�пɻ��յ���Ҫ������ ��
��1������﮻�ʯ������ĽӴ�������ӿ������Ӧ���ʣ���߽����ʣ�2�֣�
��2��Al3+��Fe3+��2�֣� ��3��2Fe2+��H2O2��2H+��2Fe3+��2H2O��3Ca(OH)2��2Fe3+��3Ca2+��2Fe(OH)3����
Ca(OH)2��Mg2+��Ca2+��Mg(OH)2����Ca2+��CO32-��CaCO3�� ��4�֣�
��4��Li2CO3���ܽ�����¶����߶���С����ˮϴ�ӿɼ���Li2CO3����ʧ��2�֣� ��5��Na2SO4��2�֣�
���������������1������Ӧ��ĽӴ���������Լӿ췴Ӧ���ʡ����Խ�﮻�ʯ���ϸ������Ŀ��������﮻�ʯ������ĽӴ�������ӿ������Ӧ���ʣ���߽����ʡ�
��2������Fe3+��Al3+��Fe2+��Mg2+������������ʽ��ȫ����ʱ����Һ��pH�ֱ�Ϊ3.2��4.7��9.0��11.1�������������ʯ��ʯ�Ե�����Һ��pH��6.0��6.5����ʱ����������������Fe3+��Al3+��
��3�������������ӵij���pH�ϴ����Ա������������������������������������ӣ��Լ����ڳ�ȥ��˫��ˮ����ǿ�����ԣ��������������ӣ���Ӧ�����ӷ���ʽΪ2Fe2+��H2O2��2H+��2Fe3+��2H2O��ʯ�����Na2CO3��Һ�ֱܷ��������ӡ�þ�����Լ������ӽ���γɳ�������ȥ����Ӧ�����ӷ���ʽ�ֱ���3Ca(OH)2��2Fe3+��3Ca2+��2Fe(OH)3����Ca(OH)2��Mg2+��Ca2+��Mg(OH)2��Ca2+��CO32-��CaCO3����
��4�������ܽ�ȱ���֪��̼��﮵��ܽ�����¶ȵ����߶����ͣ��������ˮϴ�ӿɼ���Li2CO3����ʧ��
��5������ת��ͼ��֪��������̼��﮵�ͬʱ���������������ɣ����Դ���Һc�пɻ��յ���Ҫ������Na2SO4��
���㣺����̼����Ʊ�����ͼ���й��жϡ������Լ�ʵ����������۵�

 ��У����ϵ�д�
��У����ϵ�д�����ĽṹΪCH3��CH2��COOH,�������ǰ�ȫ��Ч�ķ�ù��������,һ���Լ�ʽ̼��пΪԭ�ϵ�����������������: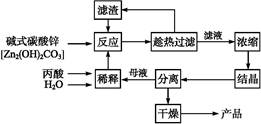
| ��� | n(����)�� n(��ʽ̼��п) | ��Ӧ�¶�/�� | ����п����/% |
| 1 | 1��0.25 | 60 | 67.2 |
| 2 | 1��0.25 | 80 | 83.5 |
| 3 | 1��0.25 | 100 | 81.4 |
| 4 | 1��0.31 | 60 | 89.2 |
| 5 | 1��0.31 | 80 | 90.1 |
| 6 | 1��0.31 | 100 | 88.8 |
(1)̽����ʵ������ѹ�������(���ϱ�):��Ӧʱ��2 h,��ˮ��45 g,n(����)��n(��ʽ̼��п)=1��
 ,��Ӧ�¶����������档
,��Ӧ�¶����������档 (2)�����ղ��á���·ѭ������ʽ,�������Ʊ����ռ�㡢���ʸ���,�������������������������������������������ŵ㡣
(3)ij��ʵ��ʱ,��37.0 g��������220 mLˮ��,�����������������Ż����������Ʊ�,���յñ���п49.6 g,��ô�ʵ�����п�IJ���Ϊ��������(д���������)��
����ĽṹΪCH3��CH2��COOH,�������ǰ�ȫ��Ч�ķ�ù��������,һ���Լ�ʽ̼��пΪԭ�ϵ�����������������: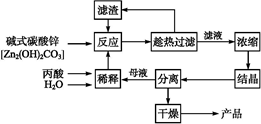
| ��� | n(����)�� n(��ʽ̼��п) | ��Ӧ�¶�/�� | ����п����/% |
| 1 | 1��0.25 | 60 | 67.2 |
| 2 | 1��0.25 | 80 | 83.5 |
| 3 | 1��0.25 | 100 | 81.4 |
| 4 | 1��0.31 | 60 | 89.2 |
| 5 | 1��0.31 | 80 | 90.1 |
| 6 | 1��0.31 | 100 | 88.8 |
(1)̽����ʵ������ѹ�������(���ϱ�):��Ӧʱ��2 h,��ˮ��45 g,n(����)��n(��ʽ̼��п)=1��
 ,��Ӧ�¶����������档
,��Ӧ�¶����������档 (2)�����ղ��á���·ѭ������ʽ,�������Ʊ����ռ�㡢���ʸ���,���������������������������ŵ㡣
(3)ij��ʵ��ʱ,��37.0 g��������220 mLˮ��,�����������������Ż����������Ʊ�,���յñ���п49.6 g,��ô�ʵ�����п�IJ���Ϊ��������(д���������)��
���������LiFePO4��һ��������������ӵ�صĵ缫���ϡ�ij�����������졢﮻�ʯLiAl��SiO3��2��������Ca2+��Mg2+���Σ���̼�۵�ԭ����������������ﮡ�����Ҫ�����������£�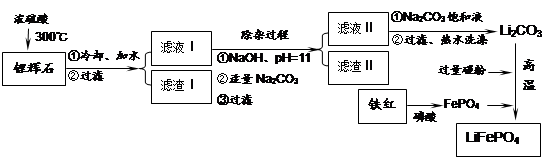
��֪��2LiAl��SiO3��2 + H2SO4(Ũ) 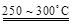 Li2SO4 + Al2O3��4SiO2��H2O��
Li2SO4 + Al2O3��4SiO2��H2O��
| �¶�/�� | 20 | 40 | 60 | 80 |
| �ܽ��(Li2CO3)/g | 1.33 | 1.17 | 1.01 | 0.85 |
| �ܽ��(Li2SO4)/g | 34.2 | 32.8 | 31.9 | 30.7 |
��1�����������пɷ����Al2O3������ͼ��ʾ����д�����ɳ��������ӷ���ʽ ��
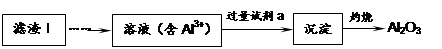
��2�����������Ҫ�ɷ��ǣ� ���ѧʽ����
��3������Һ���м��뱥��Na2CO3��Һ�����˺��á���ˮϴ�ӡ���ԭ����
��
��4��д���ڸ�����������������﮵Ļ�ѧ����ʽ ��
��5����������﮵���ܷ�ӦΪ��FePO4+Li
 LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ ��
LiFePO4������еĹ������ʿɴ���Li������д���õ�طŵ�ʱ��������Ӧ�� �����øõ�ص�ⱥ��ʳ��ˮ�����ص缫��Ϊ���Ե缫������������������4480mL���壨��״��������ʱ���õ������﮵�����Ϊ �� (1)1791�꣬����ҽ��·�������Ȼ���Ƽ�ר������ʳ�Ρ�Ũ���ᡢʯ��ʯ��úΪԭ���Ƽ�÷�����������
����NaCl��H2SO4��Ӧ��Na2SO4��2NaCl��H2SO4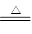 Na2SO4��2HCl��
Na2SO4��2HCl��
���ý�̿��ԭNa2SO4��Na2S��Na2SO4��4C Na2S��4CO��
Na2S��4CO��
����������ʯ��ʯ��Ӧ��Na2CO3��Na2S��CaCO3 Na2CO3��CaS
Na2CO3��CaS
(2)����˵����ȷ���� (����)��
| A���ڢ���������������ԭ��Ӧ |
| B��ֻ�еڢڲ���������ԭ��Ӧ |
| C���÷����������豸����û�и�ʴ |
| D���÷����Ի�����Ⱦ��С |