��Ŀ����
���к��ȡ���һ�����Ȼ�ѧ�е���Ҫ������к��ȡ��Ķ���Ϊ����ϡ��Һ�У��������кͷ�Ӧ������1mol H2O����ʱ�ķ�Ӧ�Ƚ����к��ȡ����¹����к��ȵ�������ȷ����( )
![]() A����ϡ��Һ��������ͼӦ���к�����ֵ�����
A����ϡ��Һ��������ͼӦ���к�����ֵ�����
![]() B����ϡ��Һ��HCl��NaOH��Ӧ��HNO3��KOH���к��ȶ����
B����ϡ��Һ��HCl��NaOH��Ӧ��HNO3��KOH���к��ȶ����
![]() C����ϡ��Һ��1mol���1mol�Ӧ�ų������������
C����ϡ��Һ��1mol���1mol�Ӧ�ų������������
![]() D����Ũ�����������������Һ�иպ�����1molˮʱ��������������Ϊ�к���
D����Ũ�����������������Һ�иպ�����1molˮʱ��������������Ϊ�к���
B

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�|
�����йط�Ӧ�ȵ�˵������ȷ���� | |
| [����] | |
A�� |
һ����ѧ��Ӧ�Ƿ����ڳ����·�����÷�Ӧ�Ħ�Hֵ�Ĵ�Сû�б�Ȼ��ϵ |
B�� |
�к��Ȧ�H����57.3��kJ��mol��1������1.00 L��1.00 mol��L��1��H2SO4��ϡ��NaOH��Һǡ����ȫ��Ӧ�ų�57.3 kJ������ |
C�� |
�õ������0.50 mol��L��1���ᡢ0.55 mol��L��1��NaOH��Һ�����к��Ȳⶨ��ʵ�飬��ʹ��õ�ֵƫ�� |
D�� |
��101 KPaʱ��1 mol��CH4��ȫȼ������CO2��ˮ�����ų�����������CH4��ȼ���� |
��15�֣�I.��9�֣�ijͬѧ��ͬ����Ԫ�����ʵݱ����ʵ��ʱ���Լ������һ��ʵ�鷽��������¼���й�ʵ���������±���
| ʵ�鷽�� | ʵ������ |
| ����ɰֽ�����þ�����ˮ��Ӧ������Ӧ����Һ�еμӷ�̪ | (A)����ˮ��,�۳�һ��С��,��ˮ���������ƶ�,��֮��ʧ,��Һ���ɫ |
| �������Ƶ�H2S������Һ�еμ����Ƶ���ˮ | (B)��������,���ڿ�����ȼ�գ���Һ���dz��ɫ |
| ��������з�̪��Һ����ˮ��Ӧ | (C)��Ӧ��ʮ��ǿ��,��������������ڿ�����ȼ�� |
| ��þ����2 mol��L��1�����ᷴӦ | (D)���ҷ�Ӧ,������ȼ������ |
| ��������2 mol��L��1�����ᷴӦ | (E) ���ɵ���ɫ���� |
���������ͬѧ���������ʵ�鱨�棺
(1)ʵ��Ŀ�ģ�̽��ͬ����Ԫ�صĽ����Ժͷǽ����Եĵݱ���ɡ�
(2)ʵ����Ʒ��
�������� �Թ� �� �ƾ��� �� ��ͷ�ι� �� �Թܼ� �� ���� �� С�� �� ����Ƭ�� ɰֽ �� �ձ��ȡ�
ҩƷ���ơ�þ����������2 mol��L��1�����ᡢ���Ƶ���ˮ�����͵�H2S��Һ�ȡ�
(3)ʵ�����ݣ�����д��ʵ�鷽�����Ӧ��ʵ������
�� �� �� �� �� ����A-E��ʾ��
д���۵����ӷ���ʽ ��
(4)ʵ����ۣ� ��
II. ��6�֣�ʵ����������ͼװ�ý����к��ȵIJⶨ����ش��������⣺
��1����ʵ��װ���Ͽ���ͼ����ȱ��һ�ֲ��������� ��
��2����1���������к��Ȳⶨʵ�飬�¶ȼ���ʹ�� �Ρ�
��3��ijͬѧΪ��ʡȥ��ϴ�¶ȼƵ��鷳������ʵ��ʱʹ����֧�¶ȼƷֱ������ͼ���¶ȣ����Ƿ�ͬ���ͬѧ�Ĺ۵㣬Ϊʲô��
��15�֣�I.��9�֣�ijͬѧ��ͬ����Ԫ�����ʵݱ����ʵ��ʱ���Լ������һ��ʵ�鷽��������¼���й�ʵ���������±���
| ʵ�鷽�� | ʵ������ |
| ����ɰֽ�����þ�����ˮ��Ӧ������Ӧ����Һ�еμӷ�̪ | (A)����ˮ��,�۳�һ��С��,��ˮ���������ƶ�,��֮��ʧ,��Һ���ɫ |
| �������Ƶ�H2S������Һ�еμ����Ƶ���ˮ | (B)��������,���ڿ�����ȼ�գ���Һ���dz��ɫ |
| ��������з�̪��Һ����ˮ��Ӧ | (C)��Ӧ��ʮ��ǿ��,��������������ڿ�����ȼ�� |
| ��þ����2 mol��L��1�����ᷴӦ | (D)���ҷ�Ӧ,������ȼ������ |
| ��������2 mol��L��1�����ᷴӦ | (E) ���ɵ���ɫ���� |
(1)ʵ��Ŀ�ģ�̽��ͬ����Ԫ�صĽ����Ժͷǽ����Եĵݱ���ɡ�
(2)ʵ����Ʒ��
���������Թ� �ھƾ��� �۽�ͷ�ι� ���ԹܼТ����Ӣ�С���߲���Ƭ��ɰֽ ���ձ��ȡ�
ҩƷ���ơ�þ����������2 mol��L��1�����ᡢ���Ƶ���ˮ�����͵�H2S��Һ�ȡ�
(3)ʵ�����ݣ�����д��ʵ�鷽�����Ӧ��ʵ������
�� �� �� �� �� ����A-E��ʾ��
д���۵����ӷ���ʽ ��
(4)ʵ����ۣ� ��
II. ��6�֣�ʵ����������ͼװ�ý����к��ȵIJⶨ����ش��������⣺

��1����ʵ��װ���Ͽ���ͼ����ȱ��һ�ֲ��������� ��
��2����1���������к��Ȳⶨʵ�飬�¶ȼ���ʹ�� �Ρ�
��3��ijͬѧΪ��ʡȥ��ϴ�¶ȼƵ��鷳������ʵ��ʱʹ����֧�¶ȼƷֱ������ͼ���¶ȣ����Ƿ�ͬ���ͬѧ�Ĺ۵㣬Ϊʲô��
��13�֣��к��ȵIJⶨ�Ǹ�����Ҫ�Ķ���ʵ�顣ȡ0.55mol/L��NaOH��Һ50mL��0.25mol/L������50mL����ͼ��ʾ��װ���н����к��ȵIJⶨʵ�飬�ش��������⣺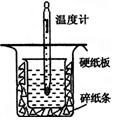
(1)����ͼʵ��װ�ÿ���������ȱ�ٵ�һ�ֲ�����Ʒ��_________ _������֮�⣬װ���е�һ�����Դ����� ��
(2)Ϊ��֤��ʵ��ɹ���ͬѧ��ȡ�������ʩ����ͼ����ֽ������������________ ___��
(3)������60mL 0.25mol��L-1 H2SO4��50mL 0.55mol��L-1 NaOH��Һ���з�Ӧ������ʵ����ȣ����ų������� �����ȡ���������ȡ�������ʵ���������ȷ���������к��� ���ȡ�������ȡ���
(4)����NaOH��Һ����ȷ�����ǣ�________�� (������ѡ��)��
A���ز������������롡 B���������������� C��һ��Ѹ�ٵ���
(5)ʹ������NaOH��Һ��Ͼ��ȵ���ȷ�����ǣ�________�� (������ѡ��)��
A�����¶ȼ�С�Ľ���
B���ҿ�ӲֽƬ�ò���������
C����������ձ�
D���������¶ȼ��ϵĻ��β���������ؽ���
(6)ʵ���������±���
������д�±��еĿհף�
| �¶� ʵ������� | ��ʼ�¶�t1�� | ��ֹ�¶�t2/�� | �¶Ȳ�ƽ��ֵ (t2��t1)/�� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 29.5 | |
| 2 | 27.0 | 27.4 | 27.2 | 32.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.2 | |
| 4 | 26.4 | 26.2 | 26.3 | 29.8 | |
������ʵ����ֵ�����57.3 kJ/mol��ƫ�����ƫ���ԭ�������(����ĸ)____ ____��
a��ʵ��װ�ñ��¡�����Ч����
b�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
c���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���