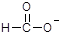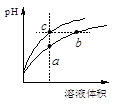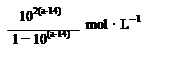��Ŀ����
25��ʱ������˵����ȷ����
A��pH =8��3��NaHCO3��Һ��c�� Na+����c�� HC ��>c��C ��>c��C  ����c�� H2CO3�� ����c�� H2CO3�� |
| B���к�ͬŨ��ͬ���������ʹ��ᣬ���ĵ�NaOH�����ʵ�������ͬ |
| C����һ��Ũ�ȵ�NaOH��Һ�ζ�����ᣬ��pH =6ʱ����Һ��c��F������c��Na+��=9��9��l0��7 mol/L |
| D����NaOH��Һ�ζ�HCI��CH3COOH�Ļ����Һ������ʱ����Һ�е��ᱻ��ȫ�к� |
C
A��pH =8��3��NaHCO3��Һ����Һ�ʼ��ԣ�˵��ˮ����ڵ��룬c�� H2CO3��>c��C  ����A����ȷ��B��ͬŨ��ͬ���������ʹ��������ʵ����ʵ�����ͬ���к�ͬŨ��ͬ���������ʹ��ᣬ���ĵ�NaOH�����ʵ�����ͬ��B����ȷ��C���ɵ���غ㣬c��F������c��Na+��=c��H�� ����c��OH�D��=10�D6�D10�D8=9��9��l0��7 mol/L,C��ȷ��D�����������ᣬ�к�������ʱ����Һ���������D����ȷ��ѡC��
����A����ȷ��B��ͬŨ��ͬ���������ʹ��������ʵ����ʵ�����ͬ���к�ͬŨ��ͬ���������ʹ��ᣬ���ĵ�NaOH�����ʵ�����ͬ��B����ȷ��C���ɵ���غ㣬c��F������c��Na+��=c��H�� ����c��OH�D��=10�D6�D10�D8=9��9��l0��7 mol/L,C��ȷ��D�����������ᣬ�к�������ʱ����Һ���������D����ȷ��ѡC��
����:�������Һ
 ����A����ȷ��B��ͬŨ��ͬ���������ʹ��������ʵ����ʵ�����ͬ���к�ͬŨ��ͬ���������ʹ��ᣬ���ĵ�NaOH�����ʵ�����ͬ��B����ȷ��C���ɵ���غ㣬c��F������c��Na+��=c��H�� ����c��OH�D��=10�D6�D10�D8=9��9��l0��7 mol/L,C��ȷ��D�����������ᣬ�к�������ʱ����Һ���������D����ȷ��ѡC��
����A����ȷ��B��ͬŨ��ͬ���������ʹ��������ʵ����ʵ�����ͬ���к�ͬŨ��ͬ���������ʹ��ᣬ���ĵ�NaOH�����ʵ�����ͬ��B����ȷ��C���ɵ���غ㣬c��F������c��Na+��=c��H�� ����c��OH�D��=10�D6�D10�D8=9��9��l0��7 mol/L,C��ȷ��D�����������ᣬ�к�������ʱ����Һ���������D����ȷ��ѡC������:�������Һ

��ϰ��ϵ�д�
 �żӾ���ϵ�д�
�żӾ���ϵ�д�
�����Ŀ