��Ŀ����
6�����ʹ���һ�ֵ�ζ�㾫�����Ǧ����¡�������ͬ���칹����ɵĻ��������ɽ��ͷ����ƷA���ṹʽ��H2O�еġ�18����Ϊ����2���ǻ�����Ϊ����ȥ�ģ�����ͼ��ʾ��Ӧ�Ƶ� �Իش�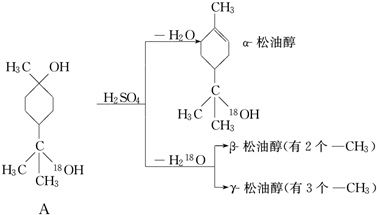
��1�������ʹ��ķ���ʽ��C10H1818O��
��2�������ʹ����������������a��a������������ b���ӡ�������c������һԪ��
��3�������ʹ����ܷ����ķ�Ӧ������ac��a���ӳɡ�������b��ˮ�⡡������c������
��4��д���ṹ��ʽ�������ʹ���
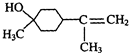 �������ʹ���
�������ʹ���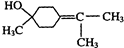 ��
��
���� ��1���������������Ħ�-���ʹ��Ľṹ��ʽ���ж�����Ԫ�ص������������д������ʽ��
��2���ɦ�-���ʹ��Ľṹ��ʽ�ɿ��������к��д��ǻ������Һ�C=C������������
��3���ɦ�-���ʹ��Ľṹ��ʽ��֪�������к���C=C�ʹ�-OH�����ݹ������жϿ��ܾ��е����ʣ�
��4������A������ȥ��Ӧ�����ʽ����ĿҪ���д��ͬ���칹��Ľṹ��ʽ��
��� �⣺��1���������������Ħ�-���ʹ��Ľṹ��ʽ����д�������ʽΪC10H1818O��Ӧע�����Ҫ��ͬλ�صķ��ű�������ʴ�Ϊ��C10H1818O��
��2���ɦ�-���ʹ��Ľṹ��ʽ�ɿ��������к��д��ǻ������Һ�C=C��������ڴ��࣬�����DZ���һԪ�����ʴ�Ϊ��a��
��3���ɦ�-���ʹ��Ľṹ��ʽ��֪�������к���C=C�ʹ�-OH���ɷ���ȡ�����ӳɺ�������Ӧ�������ܷ���ˮ�ⷴӦ���ʴ�Ϊ��ac��
��4�����ݦ�-���ʹ��к�������������д����ṹ��ʽΪ ����-���ʹ��к���������������ӵĽṹ��ʽΪ��
����-���ʹ��к���������������ӵĽṹ��ʽΪ�� ��
��
�ʴ�Ϊ�� ��
�� ��
��
���� ���⿼���˴������ʺ�ϩ���������Լ��������������Ӧ����Ŀ�Ѷ��еȣ����ڷ�Ӧ���ƶϲ��������л��⣮ֻҪ����Ū�巴Ӧ��Ľṹ�����շ�Ӧ���й����ŵ����ʣ����ݷ�Ӧ�������е�������ȥ�ƶϲ�������࣬�����ͻ�ܿ�ó���ȷ�Ĵ𰸣�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 13Al | B�� | 13Al | C�� | 13C | D�� | 13C |
| A�� | ������Ԫ���⣬�ǽ���Ԫ��ȫ����������Ԫ�� | |
| B�� | �������ڷǽ���Ԫ�غ���������Դ�����������ǿ | |
| C�� | ͬ���ڢ�A�����A��Ԫ�ص�ԭ������֮�����Ϊ25 | |
| D�� | ͬ��������������Ԫ�ص�ԭ������֮�����Ϊ32 |
| A�� | 0.0225 mol/��L•min�� | B�� | 0.05 mol/��L•min�� | ||
| C�� | 0.045 mol/��L•min�� | D�� | 0.01 mol/��L•min�� |
| A�� | Cu | B�� | CuO | C�� | CaCO3 | D�� | NaHCO3 |
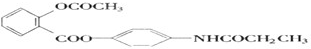 ��������ȫˮ��ʱ���õ�4���л�������ǵĽṹ��ʽ�ֱ�Ϊ
��������ȫˮ��ʱ���õ�4���л�������ǵĽṹ��ʽ�ֱ�Ϊ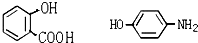 ��CH3COOH��CH3CH2OH��
��CH3COOH��CH3CH2OH��