��Ŀ����
��1�� ����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע����֪��
2Cu(s)+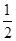 O2(g) ="==" Cu2O(s)
��H=��169kJ��mol-1��
O2(g) ="==" Cu2O(s)
��H=��169kJ��mol-1��
C(s)+ 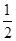 O2(g) ="==" CO(g)
��H=��110.5kJ��mol-1��
O2(g) ="==" CO(g)
��H=��110.5kJ��mol-1��
2Cu(s)+ O2(g)===2 CuO(s) ��H=��314kJ��mol-1
��ҵ����̿���ڸ��������»�ԭCuO��ȡCu2O��CO���Ȼ�ѧ����ʽΪ
��
��2��ij��ȤС���ͬѧ����ͼ��ʾװ���о��йص绯ѧ������(�ס��ҡ�����������������)�����պϸ�װ�õĵ��Kʱ���۲쵽�����Ƶ�ָ�뷢����ƫת��
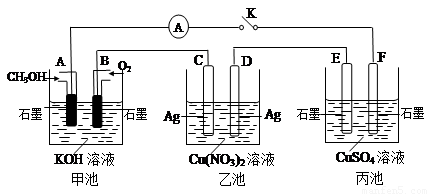
��ش��������⣺
��1���׳�Ϊ (�ԭ��ء��������ء��� ����Ƴء�)��A�缫�ĵ缫��ӦʽΪ ��
��2��������F�缫Ϊ (�����������������������������������)���óص��ܷ�Ӧ����ʽΪ ��
��3��������C����������10.8 gʱ���׳���B�缫����������O2�����Ϊ mL(��״��)��
��4��һ��ʱ��Ͽ����K������������ʹ�ҳػָ�����ӦǰŨ�ȵ��� (��ѡ����ĸ)��
A��Cu B��CuO C��Cu(OH)2 D��Cu2(OH)2CO3
��1��C(s) + 2CuO (s)= Cu2O(s) + CO(g)  H =+34.5kJ��mol.-1��3�֣�
H =+34.5kJ��mol.-1��3�֣�
��2����ԭ��� ��1�֣� CH3OH + 8OH����6e�� = CO32��+ 6H2O��2�֣�
��������1�֣� 2CuSO4+ 2H2O 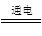 2H2SO4 + 2Cu + O2����2�֣�
2H2SO4 + 2Cu + O2����2�֣�
��560��2�֣�
��A��2�֣�
��������
�����������1����ҵ����̿���ڸ��������»�ԭCuO��ȡCu2O��CO���Ȼ�ѧ����ʽΪ
C(s) + 2CuO (s)= Cu2O(s) + CO(g)  H =a��
H =a�� H �ļ������£�C(s)+
H �ļ������£�C(s)+ 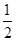 O2(g) ="==" CO(g) ��ȥ2Cu(s)+ O2(g)===2 CuO(s) �ټ���2Cu(s)+
O2(g) ="==" CO(g) ��ȥ2Cu(s)+ O2(g)===2 CuO(s) �ټ���2Cu(s)+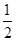 O2(g) ="==" Cu2O(s) ���Եõ�C(s) + 2CuO (s)= Cu2O(s) + CO(g) ������
O2(g) ="==" Cu2O(s) ���Եõ�C(s) + 2CuO (s)= Cu2O(s) + CO(g) ������ H
=-110.5+314-169=+34.5kJ��mol.-1�����Թ�ҵ����̿���ڸ��������»�ԭCuO��ȡCu2O��CO���Ȼ�ѧ����ʽΪ
H
=-110.5+314-169=+34.5kJ��mol.-1�����Թ�ҵ����̿���ڸ��������»�ԭCuO��ȡCu2O��CO���Ȼ�ѧ����ʽΪ
C(s) + 2CuO (s)= Cu2O(s) + CO(g)  H =+34.5kJ��mol.-1��
H =+34.5kJ��mol.-1��
��1����ͼ����֪�����׳�Ϊԭ��أ�A�缫Ϊ�������õ缫��ӦʽΪCH3OH
+ 8OH����6e�� = CO32��+ 6H2O����2��������F�缫���صĸ������ӣ�����F�缫Ϊ�����������Ϊ���أ��õ�ص��ܵķ�ӦʽΪ2CuSO4+ 2H2O 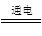 2H2SO4 + 2Cu + O2������3��C���У�Ag�ܽ⣬���Ե�����C����������10.8 gʱ����ͨ���ĵ�����Ϊ10.8/108=0.1mol������B�缫����������O2�����=0.1/4��22.4��1000=560mL����4��һ��ʱ��Ͽ����K���ҳ��ڵ��Ĺ����������������ӣ���һ����ͭ�����ڵ缫�����������Լ���Cu���Իָ���ԭ����Ũ�ȣ��ʱ���Ĵ�ѡ��A��
2H2SO4 + 2Cu + O2������3��C���У�Ag�ܽ⣬���Ե�����C����������10.8 gʱ����ͨ���ĵ�����Ϊ10.8/108=0.1mol������B�缫����������O2�����=0.1/4��22.4��1000=560mL����4��һ��ʱ��Ͽ����K���ҳ��ڵ��Ĺ����������������ӣ���һ����ͭ�����ڵ缫�����������Լ���Cu���Իָ���ԭ����Ũ�ȣ��ʱ���Ĵ�ѡ��A��
���㣺�Ȼ�ѧ����ʽ����д��ԭ��ء�����
���������⿼�����Ȼ�ѧ����ʽ����д��ԭ��ء����أ���Щ�����Ǹ߿�������ص���ѵ㣬������һ�����ۺ��ԣ��Ѷ����С�

��15�֣�����Cu2 O ��ĩ,�������ӳߴ�ЧӦ,���������Ĺ�ѧ����ѧ����绯ѧ����,��̫����ء��������������塢����͵��±�ɫ�������д����л���Ⱦ��ȷ�������DZ�ڵ�Ӧ�á�
������������ͭ���Ʊ�
��1��������ȡCu2O�ķ������£�
�ٻ�ԭ����̿���ڸ��������»�ԭCuO��
������ʵ���о����£�N2H4����ԭ����Cu(OH)2���Ʊ�����Cu2O��ͬʱ�ų�N2��
��֪��N2H4(l)+O2(g) N2(g)+2H2O(l)
��H=-a
kJ/mol
N2(g)+2H2O(l)
��H=-a
kJ/mol
Cu(OH)2(s) CuO(s)+H2O(l) ��H=b kJ/mol
CuO(s)+H2O(l) ��H=b kJ/mol
4CuO(s) 2Cu2O(s)+O2(g)
��H=c
kJ/mol
2Cu2O(s)+O2(g)
��H=c
kJ/mol
��÷����Ʊ�Cu2O���Ȼ�ѧ����ʽΪ ��
�۹�ҵ����Ҫ���õ�ⷨ����ͭ�������缫������Ȼ��ƺ��������ƵĻ����Һ������ܷ���ʽΪ��2Cu+H2O Cu2O+H2������������ӦʽΪ��
��
Cu2O+H2������������ӦʽΪ��
��
�ܻ��ɲ���Na2SO3��ԭCuSO4������Na2SO3 ��CuSO4�����ܽ���У��Ƴ�һ��Ũ�ȵ���Һ��ͨ���������ȣ���100��~104��䷴Ӧ�����Ƶá�д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
������������ͭ��Ӧ��
��2�����Ƶõ�Cu2O���д��ֽ�ˮ��ʵ��
��һ���¶��£���2 L�ܱ������м�������Cu2O��ͨ��10. 0 molˮ������������Ӧ��
2H2O(g)  2H2(g)��O2(g)
��H����484 kJ��mol��1
2H2(g)��O2(g)
��H����484 kJ��mol��1
T1�¶��²�ͬʱ�β���O2�������±���
|
ʱ��/min |
20 |
40 |
60 |
80 |
|
n(O2)/mol |
1.0 |
1.6 |
2.0 |
2.0 |
ǰ20 min�ķ�Ӧ���� v(H2O)�� ���ø��¶��£���Ӧ��ƽ�ⳣ���ı���ʽK�� ����T2�¶���K��0.4��T1 T2����>��<��=��
����ͼ��ʾ��t1ʱ�̴ﵽƽ���ֻ�ı�һ�������ִﵽƽ��IJ�ͬʱ���ڣ�H2��Ũ����ʱ��仯���������t1ʱƽ����ƶ�����Ϊ ��t2ʱ�ı����������Ϊ ������K1��K2��K3�ֱ��ʾt1ʱ����ı�����������ʱ����ڵ�ƽ�ⳣ����t3ʱ��û�м���������ϵ�е��κ����ʣ���K1��K2��K3�Ĺ�ϵΪ ��

�����������ַ����Ƶõ�Cu2O������������ͬ�·ֱ��ˮ���ֽ⣬��������������v��ʱ��t�仯��ͼ��ʾ������������ȷ���� ��

A�������ۡ����Ƶõ�Cu2O��Ч����Խϸ�
B���������Ƶõ�Cu2O������ʱ��ˮ��ƽ��ת�������
C����Ч����Cu2O�����Ĵ�ϸ��������Ե���
D��Cu2O��ˮ�ֽ�ʱ����Ҫ���˵��¶�
��1�� ����Cu2O���ھ��������Ĵ����ܶ��ܵ���ע����֪��
2Cu(s)+![]() O2(g) === Cu2O(s) ��H=��169kJ��mol-1��
O2(g) === Cu2O(s) ��H=��169kJ��mol-1��
C(s)+ ![]() O2(g) === CO(g) ��H=��110.5kJ��mol-1��
O2(g) === CO(g) ��H=��110.5kJ��mol-1��
2Cu(s)+ O2(g)=== CuO(s) ��H=��314kJ��mol-1
��ҵ����̿���ڸ��������»�ԭCuO��ȡCu2O��CO���Ȼ�ѧ����ʽΪ
��
��2��ij��ȤС���ͬѧ����ͼ��ʾװ���о��йص绯ѧ������(�ס��ҡ�����������������)�����պϸ�װ�õĵ��Kʱ���۲쵽�����Ƶ�ָ�뷢����ƫת��
 |
��ش��������⣺
��1���׳�Ϊ (�ԭ��ء��������ء��� ����Ƴء�)��A�缫�ĵ缫��ӦʽΪ ��
��2��������F�缫Ϊ (�����������������������������������)���óص��ܷ�Ӧ����ʽΪ
��
��3��������C����������10.8 gʱ���׳���B�缫����������O2�����Ϊ mL(��״��)��
��4��һ��ʱ��Ͽ����K������������ʹ�ҳػָ�����ӦǰŨ�ȵ��� (��ѡ����ĸ)��
A��Cu B��CuO C��Cu(OH)2 D��Cu2(OH)2CO3
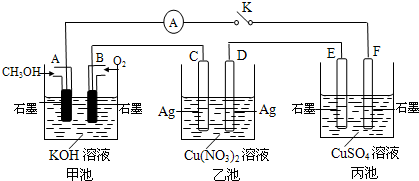
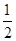 O2(g) ="==" Cu2O(s) ��H=��169kJ��mol-1��
O2(g) ="==" Cu2O(s) ��H=��169kJ��mol-1��