��Ŀ����
Ԫ�����ڱ���ӳ��Ԫ�����ʵ������Ա仯���ɣ�����ݶ�����Ԫ�������ڱ��е�λ�á��ṹ��������ʲ�����ṩ�ı���ͼ���ش��������⣺�⻯�� | �е� |
A | 1317 |
B | 100 |
C | 19.5 |
D | -33 |
E | -162 |

(1)������Ԫ���У�ԭ�ӵ���������������Ӳ�����ȵ�Ԫ����__________�֣�
(2)�ڶ�����Ԫ���У���Be��B��Ne����Ԫ���⣬����Ԫ�ص��⻯��е����ϱ���ʾ����A�ľ���������__________��C��D������Ӧ�Ļ�ѧ����ʽΪ_________________________��
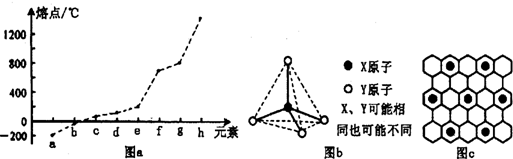
(3)��������Ԫ�ص��ʵ��۵㰴�ɵ͵���������ͼ����ʾ����gԪ�ص�����Ϊ__________��д����ҵ��ұ��h�Ļ�ѧ����ʽ______________________________��
(4)���Ӽ������Һ�����ȵĵ��������������ҵ�����ṹ��ͼ����ʾ��д���������ڼ��з�����Ӧ�����ӷ���ʽ_______________________________��
(1)3
(2)���Ӿ��� HF+NH3![]() NH
NH
(3)�� SiO2+![]() 2CO��+Si
2CO��+Si
(4)![]() +H2O
+H2O![]() NH3��H2O+H+
NH3��H2O+H+
�������������ۺϿ������ʽṹ��Ԫ�������ɡ����ڱ��й�֪ʶ��������Ը�ϰһЩ��ѧ���ɡ����۷е�Ƚϡ�

 ��Ȥ����¹�֪��ϵ�д�
��Ȥ����¹�֪��ϵ�д� Ӣ��СӢ������Ĭдϵ�д�
Ӣ��СӢ������Ĭдϵ�д� �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д���1��������Ԫ���У�ԭ�������������͵��Ӳ�����ȵ�Ԫ����________�֡�
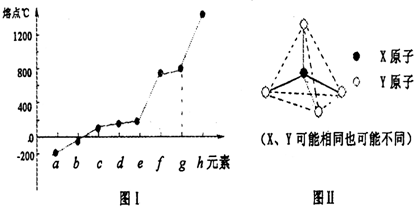
��2���ڶ�����Ԫ���У���Be��B��Ne����Ԫ���⣬����Ԫ�ص��⻯��е����±�1��ʾ������C�Ļ�ѧʽΪ________________����������Ԫ�ص��ʵ��۵㣨�棩�仯����ͼ����ʾ������h��Ԫ�ط���Ϊ________________��
�⻯�� | �е�/�� |
A | 1 317 |
B | 100 |
C | 19.5 |
D | -33 |
E | -162 |
��1

ͼ��
��3��ԭ�Ӱ뾶��С��Ԫ����ڶ����ڵ�����Ԫ�ؿɷֱ��γɷ��Ӽ������ҡ����Ӽ������Һ�����ȵĵ����������ҵ�����ṹ��ͼ����ʾ����д���ס��ҷ�Ӧ�����ӷ���ʽ_________________��

��X��Y������ͬҲ���ܲ�ͬ��
ͼ��
��4���ڶ����������ڵ�ijЩԪ�������γɾ���ͼ����ʾ�Ľṹ��Ԫ�ĵ��ʱ��ͻ����ﶡ�����Ͷ��ľ���������ͬ���ұ��붡�ڸ���ʱ�ܷ����û���Ӧ�����÷�Ӧ�б����ƻ���1 mol���ۼ�����μӷ�Ӧ�Ķ�������Ϊ__________g��