��Ŀ����
��08��ɽ��������16�֣�ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϡ�
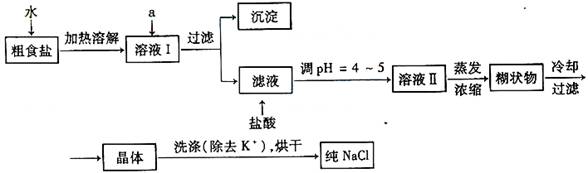
�Ŵ�ʳ�γ���������K+��Ca2+��Mg2+��Fe3+��SO42-���������ӣ�ʵ�����ᴿNaCl���������£�
�ṩ���Լ�������Na2CO3��Һ ����K2CO3��Һ NaOH��Һ BaCl2��Һ Ba(NO3)2��Һ 75%�Ҵ� ���Ȼ�̼
������ȥ��Һ���е�Ca2+��Mg2+��Fe3+��SO42-���ӣ�ѡ��a���������Լ������μ�˳������Ϊ _____ ��ֻ�ѧʽ����
��ϴ�ӳ�ȥNaCl������渽��������KCl��ѡ�õ��Լ�Ϊ_____ ��
 �����ᴿ��NaCl����500mL4.00 mol?L-1NaCl��Һ������������ҩ�ס���������� ______ �����������ƣ���
�����ᴿ��NaCl����500mL4.00 mol?L-1NaCl��Һ������������ҩ�ס���������� ______ �����������ƣ���
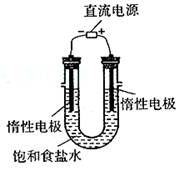
�ǵ�ⱥ��ʳ��ˮ��װ����ͼ��ʾ�����ռ���H2Ϊ2L����ͬ���������ռ���Cl2 (�����������������)2L��ԭ���� _____ ��װ�øĽ��������Ʊ�NaOH��Һ�����ⶨ��Һ��NaOH��Ũ�ȣ����õķ���Ϊ ________ ��
��ʵ�����Ʊ�H2��Cl2ͨ���������з�Ӧ��
Zn��H2SO4====ZnSO4��H2��
MnO2��4HCl(Ũ)![]() MnCl2��Cl2����2H2O
MnCl2��Cl2����2H2O
�ݴˣ���������������װ����ѡ���Ʊ����ռ�H2��װ��_____ ������ţ����Ʊ����ռ��������Cl2��װ��____ ������ţ���
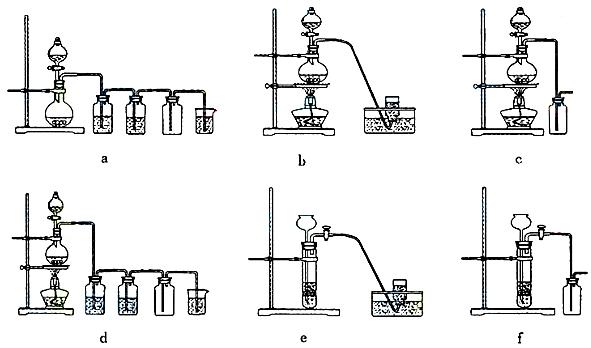
��ѡ���Ʊ������װ�ã�
��������1����Ҫ��ȥSO42����ֻ��ѡBaCl2��Һ����ѡ��Ba(NO3)2���������µ�����NO3������ѡ��NaOH��Һ��ȥMg2����Fe3����Һ�����ѡ��Na2CO3��Һ��ȥCa2�����˴�����ѡ��K2CO3��Һ������������µ�K��������HCl��ȥ������CO32����Na2CO3��Һ���ܼ���BaCl2��Һǰ�����������Ba2�����ڳ�ȥNaCl��������������KCl��Ӧѡ��75%���Ҵ�����ΪCCl4�ж���ͬʱKClҲ�����ܽ���CCl4�С�
(3��2NaCl��2H2O![]() 2NaOH��Cl2����H2�������������ڲ�����Cl2����������ɵ�NaOH��ӦNaCl��NaClO��H2O��ʹ�ò��ֵ�Cl2�����ģ�����ͬ���������ռ�����Cl2С��2L��
2NaOH��Cl2����H2�������������ڲ�����Cl2����������ɵ�NaOH��ӦNaCl��NaClO��H2O��ʹ�ò��ֵ�Cl2�����ģ�����ͬ���������ռ�����Cl2С��2L��
�𰸣�(1)��BaCl2��NaOH��Na2CO3����75���Ҵ���
(2)��ƽ���ձ���500 mL����ƿ����ͷ�ιܡ�
(3)<��������ɵ������������ɵ�NaOH�����˷�Ӧ���к͵ζ���
(4)e��d��

��08ɽ���߿���̾�����8�֣�2007��10��������ά�Ƽ����140������1867���ڰ������粩�����ϣ��������ʽ����Ϊ����ά�Ƽ���÷��㷺Ϊ����������ã����й��ĺ����Ƽ��ȡ��������ά�������������ǣ�
 |
����ά����ʵ��������������ʳ��������ֻ��70���Ҹ���ƷCaCl2û����;����Ⱦ������
1940���ҹ���ѧ�Һ�°������������ʵ�飬�����ˡ�����ά�����ļ��������������ԸĽ���ʳ�������ʴ�96���õ��˴�����Ȼ�����ֲ�Ʒ������Ϊ�������Ƽ���������ԭ���ǣ�
����30�桫50��ı���ʳ��ˮ�У���ͨ�백���ͣ���ͨ��CO2��̼�����Ƴ�����
�ڹ��ˣ����������ȶ��ø���Ʒ��
������Һ�м���ϸʳ��ĩ����10�桫15�棬ʹNH4Cl���������ˣ���ҺΪ����ʳ��ˮ��
������������⣺
��1���ڰ��ı���NaCl��Һ��ͨ��CO2��������Ӧ����ʽ��
______ ___________��
��2��������NaCl��Һ��ͨ��CO2��NaHCO3��ԭ����____________________��
��3������С�մ��ĸҺ�м�����ʯ�ҵķ�Ӧ����ʽ��_______________��
��4���ڡ������Ƽ���У�_______���ʿ���ѭ�����ã��ڡ�����ά������___________���ʿ���ѭ�����á�
��5���������Ƽ���롰����ά����ȣ����ŵ���___________��