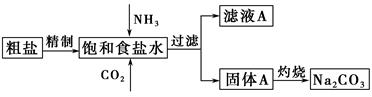
��Ŀ����
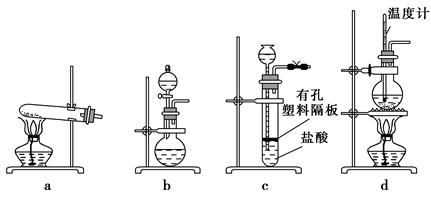
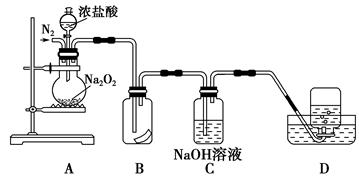
�ں�����ߺ�DZˮͧ�п���Na2O2��Ϊ����������ѡ���ʵ��Ļ�ѧ�Լ���ʵ����Ʒ������ͼ�е�ʵ��װ�ý������飬֤��Na2O2������������
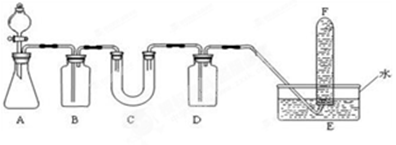
��1��A����ȡCO2��װ�á���ʵ�����CO2����ϡ������ʯ��ʯ��Ӧ����ȡ����Ӧ�Ļ�ѧ����ʽΪ�� ����2�֣�
��2����д���пո�
��3��Na2O2��CO2��Ӧ�Ļ�ѧ����ʽ�� ����2�֣�
��4���Թ�F���ռ����������һ��������ǣ� ����2�֣�

��1��A����ȡCO2��װ�á���ʵ�����CO2����ϡ������ʯ��ʯ��Ӧ����ȡ����Ӧ�Ļ�ѧ����ʽΪ�� ����2�֣�
��2����д���пո�
| ���� | �����Լ� | ������Լ���Ŀ�� |
| B | ����NaHCO3��Һ | ��2�֣� |
| C | Na2O2 | ��2�֣� |
| D | NaOH��Һ | ��2�֣� |
��3��Na2O2��CO2��Ӧ�Ļ�ѧ����ʽ�� ����2�֣�
��4���Թ�F���ռ����������һ��������ǣ� ����2�֣�
��12�֣���1��CaCO3��2HCl��CaCl2��H2O��CO2��(2��)
��2����ȥCO2�����л����HCl(2��)����CO2��ˮ��Ӧ������O2(2��)��
����δ��Ӧ��CO2����(2��)��3��2Na2O2��2CO2��2Na2CO3��O2��(2��)
��4����E�еĵ����Ƴ�ˮ�棬�رշ�Һ©����������Ĵָ��ס�Թܿڣ�ȡ���Թܣ��Ѵ����ǵ�ľ�������Թܿڡ���2�֣�
��2����ȥCO2�����л����HCl(2��)����CO2��ˮ��Ӧ������O2(2��)��
����δ��Ӧ��CO2����(2��)��3��2Na2O2��2CO2��2Na2CO3��O2��(2��)
��4����E�еĵ����Ƴ�ˮ�棬�رշ�Һ©����������Ĵָ��ס�Թܿڣ�ȡ���Թܣ��Ѵ����ǵ�ľ�������Թܿڡ���2�֣�
�����������ʵ��װ�ÿ�֪����ʵ��������CaCO3�����ᷴӦ����CO2����������ͨ������NaHCO3��Һ���Գ�ȥCO2�����л����HCl��Ȼ�����������CO2��ˮ����Ӧ����O2������ˮ���ռ�O2�����ȡ���Թܣ������Ѵ����ǵ�ľ�������Թܿ��ڣ�ľ����ȼ��֤���Թ����ռ���������������
��1��AΪ��CaCO3�����ᷴӦ����CO2����ѧ����ʽΪCaCO3+2HCl��CaCl2+H2O+CO2����
��2����������ͨ������NaHCO3��Һ���Գ�ȥCO2�����л����HCl��Ȼ�����������CO2��ˮ����Ӧ����O2���������ɵ������л��ж�����̼���壬Ӧ��NaOH��Һϴ��������δ��Ӧ��CO2���壬����ˮ���ռ�O2���ʴ�Ϊ��
| ���� | �����Լ� | ������Լ���Ŀ�� |
| B | ����NaHCO3��Һ | ��ȥCO2�����л����HCl |
| C | �������� | ��CO2��ˮ����Ӧ����O2 |
| D | NaOH��Һ | ����δ��Ӧ��CO2���� |
��3�����������������̼��Ӧ����̼���ƺ���������Ӧ�Ļ�ѧ����ʽΪ2Na2O2+2CO2��2Na2CO3+O2��
��4����ʵ���Ŀ��Ϊ֤���������ƿ������������ռ������Ҫ��֤�Ƿ�Ϊ������Ϊ��ֹ������Ӧ�Ȱ�E�еĵ����Ƴ�ˮ�棬Ȼ��رշ�Һ©���������ô����ǵ�ľ�������Թܿ��ڣ�ľ����ȼ��֤���Թ����ռ���������������

��ϰ��ϵ�д�
 ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д� A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�
�����Ŀ
 Na2O2��
Na2O2��