��Ŀ����
9��X��Y��Z��W��Ԫ�����ڱ�ǰ�����ڳ���������Ԫ�أ�ԭ��������������X�ĵ��ʼ��ЦҼ����Цм�����X�ĵ�һ�����ܱ�������Ԫ�ض���Y�ĵ縺�Ա�X��С�����̬ԭ��������p�����Ǹò�s���ӵ�������Z��ǰ36��Ԫ����ԭ�Ӱ뾶��������Ԫ�أ�Wλ�����ڱ��ĵ�8�У���1��Zλ��Ԫ�����ڱ��������ڵڢ�A�壬��̬W2+�ĺ�������Ų�ʽ��1s22s22p63s23p63d6��
��2��X��������XO2��H2O�Ƚϣ��۵�ϸߵ���H2O���ѧʽ����Z���⻯���������Ӿ��壬Z2O2�Ļ�ѧ��������AC�����ţ���
A�����Ӽ���������B�����Թ��ۼ� C���Ǽ��Թ��ۼ�D��������
��3�����������£�Y�ĵ�����������X���������������Ӧ��ˮ�����Ũ��Һ��Ӧ������Y����ۺ����ᣬд���˷�Ӧ�Ļ�ѧ����ʽS+6HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$H2SO4+6NO2��+2H2O��
��4����25�棬101kPa�£�W�ĵ�����������ȼ�պ�ָ���ԭ�¶Ⱥ�ѹǿ��ƽ��ÿת��1mol���ӷų�QkJ����������W����ȼ�յ��Ȼ�ѧ����ʽ��3Fe��s��+2O2��g��=Fe3O4��s����H=-8Q KJ/mol��
���� X��Y��Z��W��Ԫ�����ڱ�ǰ�����ڳ���������Ԫ�أ�ԭ��������������X�ĵ��ʼ��ЦҼ����Цм����䵥�ʺ���˫����������Ϊ�ǽ���Ԫ�أ���X�ĵ�һ�����ܱ�������Ԫ�ض�����Χ�����Ų�Ϊns2np3����XΪ��Ԫ�أ�Z�Ƕ�������ԭ�Ӱ뾶��������Ԫ�أ���ZΪNaԪ�أ�Wλ�����ڱ��ĵ�8�У���WΪFeԪ�أ�Y��̬ԭ��������p�����Ǹò�s���ӵ���������Χ�����Ų�Ϊns2np4��Y�ĵ縺�Ա�X��С��Y��������X����ͬһ���ڣ�ֻ�ܴ��ڵ������ڣ���YΪSԪ�أ��ݴ˽��
��� �⣺X��Y��Z��W��Ԫ�����ڱ�ǰ�����ڳ���������Ԫ�أ�ԭ��������������X�ĵ��ʼ��ЦҼ����Цм����䵥�ʺ���˫����������Ϊ�ǽ���Ԫ�أ���X�ĵ�һ�����ܱ�������Ԫ�ض�����Χ�����Ų�Ϊns2np3����XΪ��Ԫ�أ�Z�Ƕ�������ԭ�Ӱ뾶��������Ԫ�أ���ZΪNaԪ�أ�Wλ�����ڱ��ĵ�8�У���WΪFeԪ�أ�Y��̬ԭ��������p�����Ǹò�s���ӵ���������Χ�����Ų�Ϊns2np4��Y�ĵ縺�Ա�X��С��Y��������X����ͬһ���ڣ�ֻ�ܴ��ڵ������ڣ���YΪSԪ�أ�
��1��Z��NaԪ��λ��Ԫ�����ڱ��������ڵڢ�A�壬��̬W2+���������ӣ����������Ų�ʽ��1s22s22p63s23p63d6��
�ʴ�Ϊ��������A��1s22s22p63s23p63d6��
��2��N��������NO2��H2O�Ƚϣ���Ϊˮ���Ӽ�������������ˮ���۵�ߣ�ZΪNaԪ�أ����⻯����NaH�����Ӿ��壬���������д������Ӽ��ͷǼ��Թ��ۼ���
�ʴ�Ϊ��H2O�����ӣ�AC��
��3�����������£�S����������HNO3Ũ��Һ��Ӧ������H2SO4��HNO3����ԭΪNO2����Ӧ����ʽΪ��S+6HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$H2SO4+6NO2��+2H2O��
�ʴ�Ϊ��S+6HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$H2SO4+6NO2��+2H2O��
��4����25��C��1O1kPa�£�Fe�ĵ�����������ȼ������Fe3O4���ָ���ԭ�¶Ⱥ�ѹǿ��ƽ��ÿת��1mol���ӷų�QkJ����������3moFe��Ӧ�ų�������Ϊ$\frac{3mol��\frac{8}{3}}{1mol}$��QkJ=8QkJ��Fe����ȼ�յ��Ȼ�ѧ����ʽ��3Fe��s��+2O2��g��=Fe3O4��s����H=-8Q KJ/mol��
�ʴ�Ϊ��3Fe��s��+2O2��g��=Fe3O4��s����H=-8Q KJ/mol��
���� ���⿼��ṹλ�����ʹ�ϵ����������Ų����ɡ�������ԭ��Ӧ���Ȼ�ѧ����ʽ��д�ȣ��Ѷ��еȣ��ƶ�Ԫ���ǽ���Ĺؼ���ע�����X���ʻ�ѧ����Ԫ�ص�����ȷ��XΪ��Ԫ�أ�

 ������ʱͬ����ϰ��ϵ�д�
������ʱͬ����ϰ��ϵ�д� ѧҵ����һ��һ��ϵ�д�
ѧҵ����һ��һ��ϵ�д�| A�� | H2O | B�� | CH3COOC2H5 | C�� | CH3CH2OH | D�� | CH3COOH |
| A�� | �Ҵ�������Ʒ�Ӧ | B�� | ����Һ�巴Ӧ�����屽 | ||
| C�� | ��ϩ��������Ȼ�̼��Һ��Ӧ | D�� | ������Ҵ���Ӧ������������ |
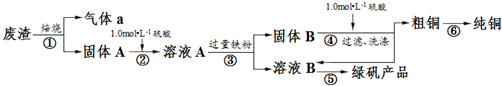
��1����ʵ�����У�����98%��Ũ���ᣨ�ܶ�Ϊ1.84g•mL-1������500mL1.0mol•L-1�����ᣬ��Ҫ����������Ͳ���ձ����������⣬���н�ͷ�ιܡ�500mL����ƿ��
��2����С��ͬѧ�������װ��ģ������ڹ��������б��գ�����֤�����к���Ԫ�أ�
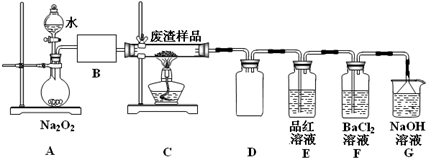
��װ��A�з�Ӧ�Ļ�ѧ����ʽΪ2Na2O2+2H2O=4NaOH+O2����Ϊ���Ʒ�Ӧ�����ڼ��Ҳ�����ƽ����������ȡ�IJ����������Ǵ�Һ©���Ͽڻ��������Ʒ�Һ©��������ʹˮ������ε��£�B��Ӧ����ʢ�м�ʯ�ҵĸ���ܣ���U�ιܣ���Ũ�����ϴ��ƿ����д�Լ����������ƣ���
��Eװ���м���Ʒ����Һ��Ŀ���Ǽ�������a�е�SO2����Fװ���г��ְ�ɫ����ʱ����Ӧ���ӷ���ʽΪ2SO2+O2+2H2O+2Ba2+=2BaSO4��+4H+��
��3�����в����У������ڲ�����н��еIJ�������ad�������и�������ţ���
��������ɴ�ͭ�õ���ͭ�ķ���Ϊ��⾫������д���ƣ���
��4��Ϊ�ⶨ��Ʒ���̷���������������ȡ30.000g��Ʒ����ˮ���250mL��Һ��ȡ25.00mL��Һ����ƿ�У���0.1000mol•L-1����KMnO4��Һ���еζ�����ӦΪ��10FeSO4+8H2SO4+2KMnO4=2MnSO4+5Fe2��SO4��3+K2SO4+8H2O��ʵ�������������±���ʾ��
| ����� | 1 | 2 | 3 | 4 |
| KMnO4��Һ���/mL | 20.90 | 20.02 | 19.98 | 20.00 |
a����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
b����ƿϴ����δ����
c���ζ��յ�ʱ���Ӷ���
d���ζ��յ�ʱ���Ӷ���
�ڸ��ݱ������ݣ��������ò�Ʒ���̷�����������Ϊ92.7%��
| A�� | �����������γɹ⻯ѧ�����������һ����Ҫԭ�� | |
| B�� | �������Ǹ��벣����ά�γɵĸ��ϲ��� | |
| C�� | ʯ�ͻ��������������ˮ�帻Ӫ���� | |
| D�� | ������dz��õ�һ����̬���� |
| A�� | ��ʳ����ϴ��ˮƿ�е�ˮ�� | |
| B�� | �����г��õľ������װ���Ǹ��ݽ������������ʶ���Ƶ� | |
| C�� | ���������������������� | |
| D�� | Ϊ���ӳ���ʵ��ij����ڣ����ý��ݹ����������Һ�Ĺ�������ˮ�����������ϩ |
| A�� | ����Һ�������ӵ�Ũ�ȣ�c��H+��=1��10-11mol•L | |
| B�� | pH=7��NH4Cl��NH3•H2O�Ļ����Һ��c��Cl-����c��NH4+����c��H+����c��H+��=c��OH-�� | |
| C�� | Ũ�Ⱦ�Ϊ0.1mol•L-1��NH3•H2O��NH4Cl��Һ�������Ϻ�ļ�����Һ�У�c��NH4+����c��Cl-����c��NH3•H2O����c��OH-����c��H+�� | |
| D�� | 0.1mol•L-1�İ�ˮ��0.05mol��H2SO4��Һ�������Ϻ�������Һ�У�2c��NH4+��+2c��NH3•H2O��=c��SO42-�� |
| A�� | 1 mol Cl2�μӷ�Ӧת�Ƶ�����һ��Ϊ2NA | |
| B�� | �ڷ�ӦKIO3+6HI�TKI+3I2+3H2O�У�ÿ����3 mol I2ת�Ƶĵ�����Ϊ6NA | |
| C�� | Na2O2��CO2��Ӧ���ɱ����11.2L O2����Ӧ������ת�Ƶ�����1 NA | |
| D�� | ��Ӧ14CuSO4+5FeS2+12H2O=7Cu2S+5FeSO4+12H2SO4�÷�ӦFeS2�е���Ԫ��ȫ�������� |
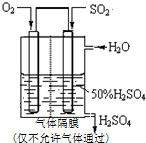 ��֪��SO2��g��+$\frac{1}{2}$O2��g��?SO3��g����H=-98kJ•mol-1��
��֪��SO2��g��+$\frac{1}{2}$O2��g��?SO3��g����H=-98kJ•mol-1��