��Ŀ����
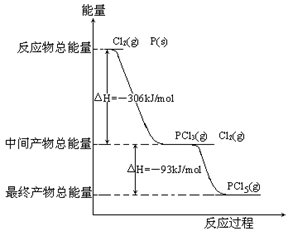 ��15�֣�����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��������ͼ�ش��������⣺
��15�֣�����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��������ͼ�ش��������⣺
(1)PCl5�ֽ��PCl3��Cl2���Ȼ�ѧ����ʽ______________�������ֽⷴӦ��һ�����淴Ӧ���¶�T1ʱ�����ܱ������м�0.80molPCl5����Ӧ�ﵽƽ��ʱPCl5��ʣ0.60mol����ֽ��ʦ�1����_________������Ӧ�¶���T1���ߵ�T2��ƽ��ʱPCl5�ķֽ���Ϊ��2����1_______��2 (����ڡ�����С�ڡ����ڡ�)��
(2)P��Cl2��������Ӧ����1molPCl5�ġ�H3��_________��P��Cl2һ����Ӧ����1molPCl5�ġ�H4______��H3(����ڡ�����С�ڡ����ڡ�)��
(1��PCl5(g)===PCl3(g)��Cl2(g) ��H��93kJ?mol��1��25����С�ڡ�
(2����399kJ?mol��1�����ڡ�

��ϰ��ϵ�д�
�����Ŀ
 ����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��
����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��


