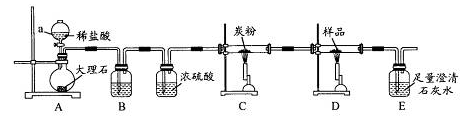
��Ŀ����
��֪���ӷ�Ӧ��Fe3����3SCN�� Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
��ȡ10 mL l mol��L��1FeCl3��Һ���μ�3��4��ŨKSCN��Һ������Һ������ɺ�
ɫ��
��ȡ������ɫ��Һ���μ�����Ũ���ᣬ���ã���Һ��ɫ��ȥ��ͬʱ���������ĺ���ɫ��
������A��
�۽�����������Aͨ�������Ba��OH��2��Һ�У�������ɫ����B��ʣ������C��
����C��ɫ��ζ����ʹȼ�յ�ľ��Ϩ�𣬿��ŷŵ������У�����ı�����ijɷ֡�
�ܹ��ˣ����ɫ����B�еμ�����ϡ���ᣬ������ȫ�ܽ⣬ͬʱ������ʹ����ʯ��ˮ��
���ǵ���ɫ��ζ����D��
��ȡ���з�Ӧ����Һ�������μ�BaCl2��Һ������������ϡ����İ�ɫ����E��
��������ʵ�����ش��������⣺
��1��B�Ļ�ѧʽΪ_________________��E�Ļ�ѧʽΪ___________________��
��2���������A�ijɷ���___________________���ѧʽ����
��3������ȤС��ͬѧ��������ʵ����������ó����ۣ�Fe��SCN��3���л�ԭ�ԣ���ʵ����з�Ӧʱ��������Ԫ����____________����Ԫ�ط��ţ�������Ԫ��C��S��N��ԭ�ӽṹ���ۼ������֪ʶ�ƶϳ�SCN���ĽṹʽΪ_________________��
��4��ʵ����з�Ӧ�����ӷ���ʽΪ___________________________________________��
��5������ȤС��ͬѧ������ʵ���еõ�����������SCN����Ӽ���Fe2��ʱӦע��_________________________________________________________________.
 Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺
Fe��SCN��3���з�ӦѸ�١��������Ե��ص㣬�Ǽ���Fe3�����õķ���֮һ��ij��ѧ��ȤС��Ϊ̽��Fe��SCN��3�����ʣ���������ʵ�飺��ȡ10 mL l mol��L��1FeCl3��Һ���μ�3��4��ŨKSCN��Һ������Һ������ɺ�
ɫ��
��ȡ������ɫ��Һ���μ�����Ũ���ᣬ���ã���Һ��ɫ��ȥ��ͬʱ���������ĺ���ɫ��
������A��
�۽�����������Aͨ�������Ba��OH��2��Һ�У�������ɫ����B��ʣ������C��
����C��ɫ��ζ����ʹȼ�յ�ľ��Ϩ�𣬿��ŷŵ������У�����ı�����ijɷ֡�
�ܹ��ˣ����ɫ����B�еμ�����ϡ���ᣬ������ȫ�ܽ⣬ͬʱ������ʹ����ʯ��ˮ��
���ǵ���ɫ��ζ����D��
��ȡ���з�Ӧ����Һ�������μ�BaCl2��Һ������������ϡ����İ�ɫ����E��
��������ʵ�����ش��������⣺
��1��B�Ļ�ѧʽΪ_________________��E�Ļ�ѧʽΪ___________________��
��2���������A�ijɷ���___________________���ѧʽ����
��3������ȤС��ͬѧ��������ʵ����������ó����ۣ�Fe��SCN��3���л�ԭ�ԣ���ʵ����з�Ӧʱ��������Ԫ����____________����Ԫ�ط��ţ�������Ԫ��C��S��N��ԭ�ӽṹ���ۼ������֪ʶ�ƶϳ�SCN���ĽṹʽΪ_________________��
��4��ʵ����з�Ӧ�����ӷ���ʽΪ___________________________________________��
��5������ȤС��ͬѧ������ʵ���еõ�����������SCN����Ӽ���Fe2��ʱӦע��_________________________________________________________________.
��1��BaCO3(1��); BaSO4(1��)
��2��NO2��CO2��N2��2�֣�
��3��S��N��2�֣���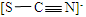 ��1�֣�
��1�֣�
��4��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��2�֣�
��5���������������ܹ�����2�֣�
��2��NO2��CO2��N2��2�֣�
��3��S��N��2�֣���
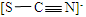 ��1�֣�
��1�֣���4��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��2�֣�
��5���������������ܹ�����2�֣�
����������ɢ��еĺ���ɫ�������A��֪A����NO2,�ɢۢܿ�֪BΪBaCO3��CΪN2��DΪCO2����A��NO2��N2��CO2��ɣ��ɢݿ�֪EΪBaSO4��2�� ��A��NO2��N2��CO2��ɣ�3���ɲ��PԪ�صĻ��ϼۿ�֪��������Ԫ����S��N������Ԫ��C��S��N��ԭ�ӽṹ���ۼ������֪ʶ�ƶϳ�SCN���ĽṹʽΪ
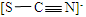 ����4�����������P�����غ�͵���غ㡢�����غ���ƽ����ʽΪ��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��5���������֪SCN-Ҳ�ܱ��������������ʼ�������������ܹ���
����4�����������P�����غ�͵���غ㡢�����غ���ƽ����ʽΪ��2SCN����22NO3����20H��=2SO42����2CO2����22NO2����N2��10H2O��5���������֪SCN-Ҳ�ܱ��������������ʼ�������������ܹ���
��ϰ��ϵ�д�
 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
�����Ŀ


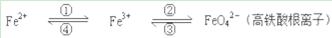

 ����ԭ��Mn2+����Ӧ�����Һ�м�������п�ۣ�����ɫ�պ���ʧ��������Һ�ռ�����ƿ�У���ʱ��Һ�Գ����ԡ�
����ԭ��Mn2+����Ӧ�����Һ�м�������п�ۣ�����ɫ�պ���ʧ��������Һ�ռ�����ƿ�У���ʱ��Һ�Գ����ԡ�