��Ŀ����
1����1������ʽΪC4H8��ϩ����3��ͬ���칹�壬��ṹ��ʽ�ֱ�Ϊ��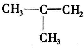 ��CH3CH=CHCH3��CH3CH2CH=CH2��
��CH3CH=CHCH3��CH3CH2CH=CH2����2��ijͬѧΪij������Ϊ��1��2-�������顱������Ϊ����ȷ������ȷ����ȷ����������ȷ����д����ȷ����2-�����飨����ȷ���ÿղ���� ��д��������ͬ���칹���к��ĸ��������Ľṹ��ʽC��CH3��4��
��3����ϩ��ʯ���ѽ����Ҫ����֮һ������ϩͨ��������Ȼ�̼��Һ�У��۲쵽����������ɫ���䷴Ӧ����ʽΪCH2=CH2+Br2��CH2BrCH2Br����ϩ��һ�������·����Ӿ۷�Ӧ�Ļ�ѧ����ʽ��
 �������������Ǿ���ϩ����ϩ��ˮ�����д��칦�ܣ�
�������������Ǿ���ϩ����ϩ��ˮ�����д��칦�ܣ�
���� ��1������ʽΪC4H8��ϩ����3��ͬ���칹�壬����̼���칹��������λ���칹�ж�ͬ���칹�壻
��2��������λ��1��̼ԭ���ϣ���������ʱѡȡ�̼��Ϊ��������֧�������̼ԭ��Ϊ1��̼ԭ�ӣ�������ͬ���칹���к��ĸ���������2��2-�������飻
��3����ϩ�к���̼̼˫�����������ʽϻ��ã��ܺ��巢���ӳɷ�Ӧ��ʹ����ɫ����ϩ��һ�������·����Ӿ۷�Ӧ���ɾ���ϩ����ϩ���д������ã�
��� �⣺��1������ʽΪC4H8��ϩ����3��ͬ���칹�壬����ϩ����̼���칹��������λ���칹֪������ͬ���칹��ṹ��ʽΪCH3CH=CHCH3��CH3CH2CH=CH2���ʴ�Ϊ��CH3CH=CHCH3�� CH3CH2CH=CH2��
��2��������λ��1��̼ԭ���ϣ����Ը����Ʋ���ȷ��Ӧ����2-�����飻��������ʱѡȡ�̼��Ϊ��������֧�������̼ԭ��Ϊ1��̼ԭ�ӣ�������2-�����飻������ͬ���칹���к��ĸ���������2��2-�������飬�ṹ��ʽΪC��CH3��4��
�ʴ�Ϊ������ȷ��2-�����飬C��CH3��4��
��3����ϩ�к���̼̼˫�����������ʽϻ��ã��ܺ��巢���ӳɷ�Ӧ��ʹ����ɫ�����Կ�������������Һ��ɫ����Ӧ����ʽΪCH2=CH2+Br2��CH2BrCH2Br����ϩ��һ�������·����Ӿ۷�Ӧ���ɾ���ϩ����Ӧ����ʽΪ ����ϩ���д������ã�
����ϩ���д������ã�
�ʴ�Ϊ����ɫ��CH2=CH2+Br2��CH2BrCH2Br�� ������ϩ�����죮
������ϩ�����죮
���� ���⿼��ͬ���칹����д��������������ϩ�����ʵ�֪ʶ�㣬���ؿ������֪ʶ���ѵ���ͬ���칹����д��ͬ���칹�����̼���칹���������칹��˳���칹��

| A�� | 0.64g | B�� | 1.28g | C�� | 2.56g | D�� | 5.12g |
| A�� | ��ˮ��pH�����¶ȵ����߶����� | B�� | ��35��ʱ��[H+]��[OH-] | ||
| C�� | ��25���35��ʱ��[H+]=[OH-] | D�� | ˮ�ĵ��������ȵ� |
| A�� | OH-+CO32-+2H+��HCO3-+H2O | B�� | 2OH-+CO32-+3H+��HCO3-+2H2O | ||
| C�� | 2OH-+CO32-+4H+��CO2��+2H2O | D�� | OH-+CO32-+3H+��CO2��+2H2O |
 ��ͬ����ǣ�������
��ͬ����ǣ�������| A�� | 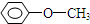 | B�� | 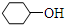 | C�� | CH3CH2OH | D�� | CH3OH |
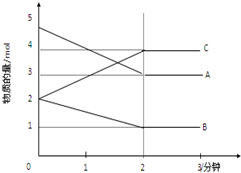 ij���淴Ӧ��ij���Ϊ5L���ܱ������н��У��ڴ�0-3���Ӹ����ʵ����ı仯�����ͼ��ʾ��A��B��C��Ϊ���壩��
ij���淴Ӧ��ij���Ϊ5L���ܱ������н��У��ڴ�0-3���Ӹ����ʵ����ı仯�����ͼ��ʾ��A��B��C��Ϊ���壩��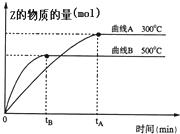 ��a L�ܱ������м���1mol X�����2mol Y���壬�������·�Ӧ��X��g��+2Y��g��?2Z��g������ͬ�¶��£����ƽ��״̬Z�����ʵ����仯������ͼ��ʾ��
��a L�ܱ������м���1mol X�����2mol Y���壬�������·�Ӧ��X��g��+2Y��g��?2Z��g������ͬ�¶��£����ƽ��״̬Z�����ʵ����仯������ͼ��ʾ��