��Ŀ����
��10�֣�W��X��Y��Z��ԭ���������������ͬһ������Ԫ�أ�W��X�ǽ���Ԫ�أ�Y��Z�Ƿǽ���Ԫ�أ�W��X���Ե�����������Ӧ��ˮ������Է�Ӧ�����κ�ˮ��
�ش��������⣺
��1��W��Y���γɻ�����W2Y���û�����ĵ���ʽΪ��__________________��
��2���Ƚ�Y��Z��̬�⻯����ȶ��ԣ�________��________���÷���ʽ��ʾ����]
��3��W��X��Y��Z����Ԫ���γɵļ����ӵ����Ӱ뾶�ɴ�С��˳���ǣ�
________��________��________��________�������ӷ��ű�ʾ����
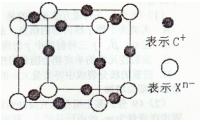
��4����ͼ��WZ�����һ���������ṹ��ʾ��ͼ��
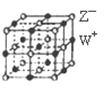
����˵����ȷ����________��
��5��Z�����������Ϊ��ɫҺ�壬0.25 mol��������һ����ˮ��ϵõ�һ ��ϡ����Һ�����ų�a KJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ__________________________________��
��ϡ����Һ�����ų�a KJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ__________________________________��
�ش��������⣺
��1��W��Y���γɻ�����W2Y���û�����ĵ���ʽΪ��__________________��
��2���Ƚ�Y��Z��̬�⻯����ȶ��ԣ�________��________���÷���ʽ��ʾ����]
��3��W��X��Y��Z����Ԫ���γɵļ����ӵ����Ӱ뾶�ɴ�С��˳���ǣ�
________��________��________��________�������ӷ��ű�ʾ����
��4����ͼ��WZ�����һ���������ṹ��ʾ��ͼ��

����˵����ȷ����________��
| A����W+ ��������������Z�� ����8�� |
| B����W+ ��������������Z�� ���ɵĶ������ǰ����� |
| C����W+ ��������������W+ ��8�� |
| D��һ��WZ�����к���4��W+ ��4��Z�� |
 ��ϡ����Һ�����ų�a KJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ__________________________________��
��ϡ����Һ�����ų�a KJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ__________________________________����10�֣�
��1��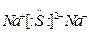
��2��HCl��H2S
��3��S2����Cl ��Na����Al3��
��4��B D
��5��Cl2O7(l)+H2O(l)��2HClO4(aq)����H����4Q kJ/mol
��1��
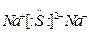
��2��HCl��H2S
��3��S2����Cl ��Na����Al3��
��4��B D
��5��Cl2O7(l)+H2O(l)��2HClO4(aq)����H����4Q kJ/mol
��

��ϰ��ϵ�д�
 �����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӽݾ��㽭��ѧ������ϵ�д�
�����Ŀ
 ��
�� ����֮�͵�������Y��Z�������ڣ�Z��Wλ��ͬһ���塣
����֮�͵�������Y��Z�������ڣ�Z��Wλ��ͬһ���塣
 ����
����

 (10��)X��Y��Z��W
(10��)X��Y��Z��W Z�ļ����Ӱ뾶(ѡ���������������������)��
Z�ļ����Ӱ뾶(ѡ���������������������)�� ����
���� ɫ����Ӧ�������漰�����ӷ���ʽΪ��
ɫ����Ӧ�������漰�����ӷ���ʽΪ��  �� ��
�� ��